Table of Contents
An electric force is an interaction between two charged bodies, either attractive or repulsive. Similar to other forces, this force effects and impacts a particular object and can easily be demonstrated by Newton’s law of motion. The electric force is one of the forces which is exerted over other bodies.
Newton’s law can also be used to analyze the effects of motion when this kind of force is present. An analysis begins by constructing a free body image in which the direction of that image and the individual force types are seen by a vector which can help calculate the total. The net force applied to a body in order to calculate acceleration is known as the net force.
What Is Electric Force?
It is the electric force that is responsible for attracting or repelling any two charged objects. In the same way as any force, its effects on objects are described by Newton’s laws of motion. With Felect, the electric force can act on objects in addition to a wide range of other forces. Newton’s laws are used to analyze the motion (or lack of motion) of objects under such a force or combination of forces.
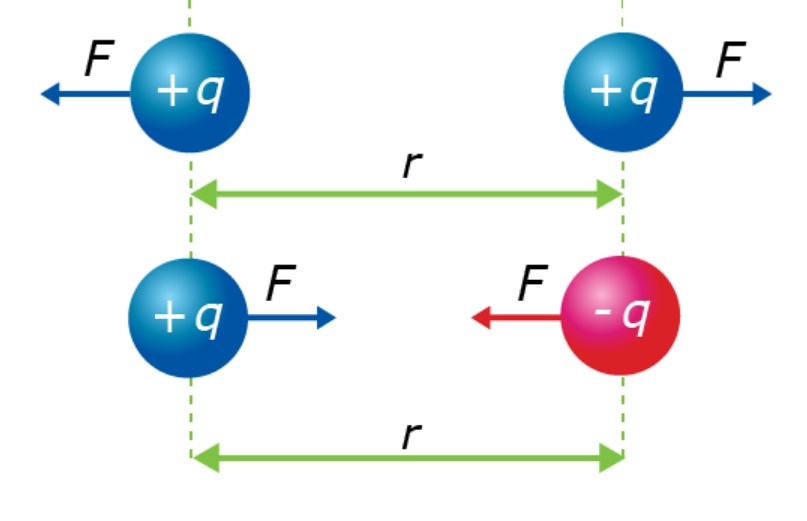
Analyses usually begin with the construction of a free-body diagram where the type and direction of forces are represented by vector arrows and labeled according to the type. As a result, the magnitudes of the forces are added as vectors to determine the resultant sum, also known as the net force. The net force can then be used to calculate the acceleration of the object.
The goal of the analysis may not always be to determine the acceleration of the object. The free-body diagram is instead used to determine the distance or charge between two objects that are at static equilibrium. In this case, a free-body diagram is used in conjunction with vector concepts in order to solve a puzzle involving geometry, trigonometry, and Coulomb’s law. This article will explore both types of applications of Newton’s laws to static electricity.
Definition
The repulsive or attractive interaction between any two charged bodies is called an electric force. Similar to any force, its impact and effects on the given body are described by Newton’s laws of motion. The electric force is among the list of other forces that exert over objects.
Newton’s laws are applicable to analyze the motion under the influence of that kind of force or combination of forces. The analysis begins with the construction of a free body image wherein the direction and type of the individual forces are shown by the vector to calculate the resultant sum which is called the net force that can be applied to determine the body’s acceleration.
Charge
How do we know there is such a thing as charge? The concept of charge arises from an observation of nature: We observe forces between objects. Electric charge is the property of objects that gives rise to this observed force. Like gravity, electric force “acts at a distance”. The idea that a force can “act at a distance” is pretty mind-blowing, but it’s what nature really does.
Electric forces are very large, far greater than the force of gravity. Unlike gravity, there are two types of electric charge, (whereas there is only one type of gravity; gravity only attracts).
Types Of Electric Force
The electric force consists of two different types of charges. They are positive electric charge and negative electric charge. The interaction of both of these charges is easily predictable. Unlike electric charges, they attract each other, while the like charges repel one another. This basically means that if the two charges are positive then there exists a repulsive force between both the charges.
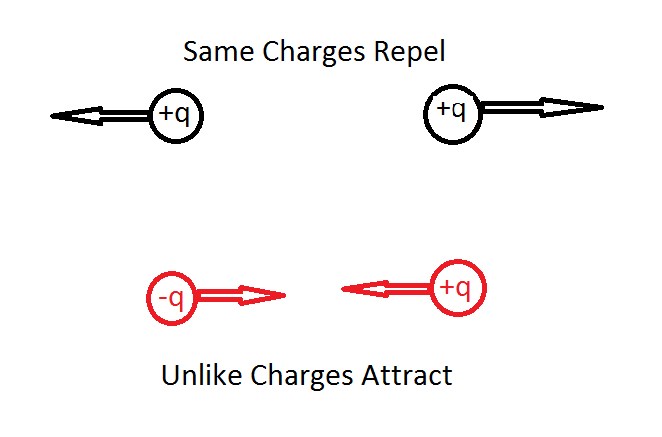
The same thing happens if two negative charges come in contact, they both will repel each other too. In contrast, if one positive charge and one negative charge come into contact, then there is generated an attractive force, in which both the charges will be attracted towards one another. The given below is the diagrammatic representation of the same.
Electric Force And Charged Particles
Electric force can happen among all the charged particles irrespective of the type of charge. These are seen as tiny particles and can be found inside atoms. They are termed protons and electrons. Protons are made up of positively charged particles whereas electrons are built by negatively charged ones. The other objects which are produced from atoms become charged due to an imbalance of the number of electrons and protons contained inside the atoms.
In an atom, protons are present inside the nucleus and are very tightly bound. They cannot move or travel across the atoms or nucleus. Meanwhile, electrons can be seen away from the atoms in their own orbitals. They can move around the atom.
What is Coulomb’s Law For Electric Force?
Coloumb’s law is an experimental law that quantifies the amount of force between two stationary electrically charged particles. The electric force between stationary charged bodies is conventionally known as the electrostatic force or Coloumb’s force. Coulomb’s law describes the amount of electrostatic force between stationary charges.
Electric Field Between Two Plates: Magnitude, Direction, Examples & More
Coulomb’s law states that:
The value of the electrostatic force of interaction between two point charges is directly proportional to the scalar multiplication of the charges and inversely proportional to the square of the distance among them.
Formula Of Coulomb’s Law For Electric Force Between Point Charges
Coulomb’s Law very nicely describes this natural phenomenon. The law has this form,
$$\vec F = K\frac{q_0.q_1}{r^2}\hat{r}$$
Where
- \(\vec{F}\) is the electric force, directed on a line between the two charged bodies.
- \(K\) is a constant of proportionality that relates the left side of the equation (newtons) to the right side (coulombs and meters). It is needed to make the answer come out right when we do a real experiment.
- \(q_0\) and \(q_1\) represent the amount of charge on each body, in units of coulombs (the SI unit for a charge).
- \(r\) is the distance between the charged bodies.
- \(\hat{r}\) is a variable unit vector that reminds us of the force points along the line between the two charges. If the charges are alike, the force is repulsive; if the charges are unlike, the force is attractive.
The Electric Constant, \(\epsilon_0\), The Permittivity Of Free Space
\(K\), the constant of proportionality, frequently appears in this form,
$$K = \frac{1}{4\pi \epsilon_0}$$
and Coulomb’s Law is written in this form,
$$\vec{F} = \frac{1}{4\pi\epsilon_0}\frac{q_0. q_1}{r^2}\hat{r}$$
The Greek letter \(\epsilon_0\) is the electric constant, also known as the permittivity of free space, (free space is a vacuum). Coulomb’s Law describes something that happens in nature. The electric constant, \(\epsilon_0\), describes the experimental setup and the system of units.
“Experimental conditions” refers to measuring \(\vec F\), with, vector, on top of point charges (or something that acts like a point charge, like charged spheres). In the SI system of units, \(\epsilon_0\) is experimentally measured to be,
$$\epsilon_0 = 8.854 187 817 \times 10^{−12}\frac{coulomb^2}{newton-meter^2}$$
This value of \(\epsilon_0\) makes,
$$K = \frac{1}{4\pi \epsilon_0} = \frac{1}{4\pi \times 8.854 \times 10^{−12} } = 8.987 \times 10^{9}$$
or for engineering purposes, we round \(K\) to something easier to remember,
$$K = \frac{1}{4\pi \epsilon_0} = 9 \times 10^{9}$$
The dimensions of \(K\) are: \(\frac{newton-meter^2}{coulomb^2}\).
Electric Force Between Line Of Charge With A Point Charge Off The End
A line of charge \(L\) meters long has a total charge of \(Q\). Assume the total charge, \(Q\), is uniformly spread out on the line. A point charge \(q\) is positioned \(a\) meters away from one end of the line.
Find the total force on a charge \(q\) positioned off the end of a line of charge.
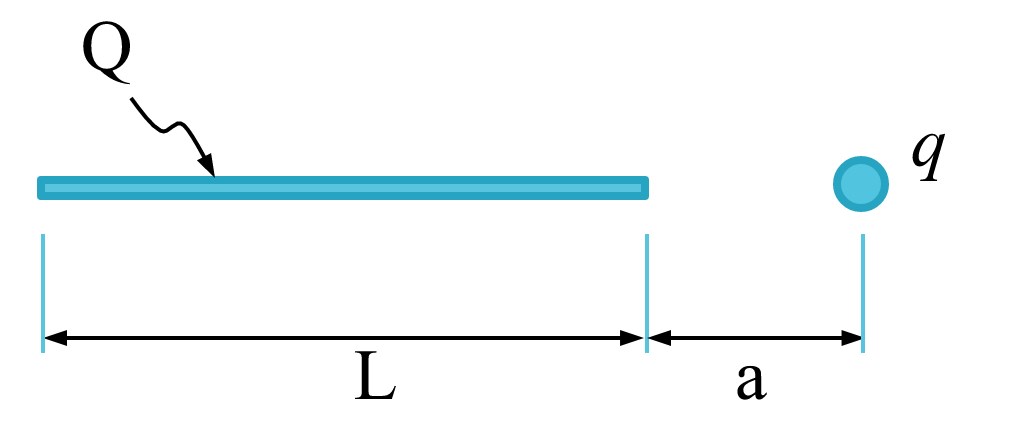
The line contains a total charge \(Q\) coulombs. We can approach this problem by thinking of the line as a bunch of individual point charges sitting shoulder to shoulder. To compute the total force on \(q\) from the line, we sum up (integrate) the individual forces from each point charge in the line.
We define the charge density in the line as \(\frac{Q}{L} \frac{coulombs}{meter}\).
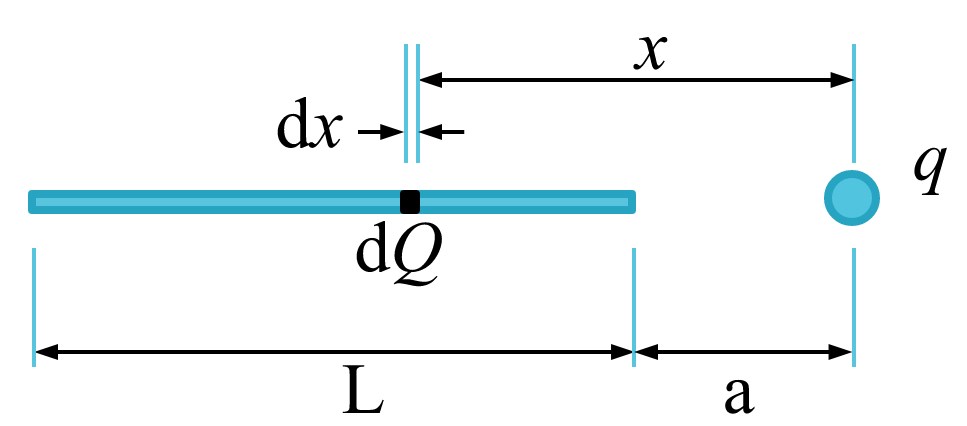
The idea of charge density lets us express the amount of charge, \(dQ\), in a little piece of the line, \(dx\), as,
\(Q\) is close enough to be a point charge to allow us to apply Coulomb’s Law. We can figure out the direction of the force right away: The force on \(q\) from every \(Q\) is directed straight between \(q\) and \(dQ\). Direction solved, now the magnitude of the force,
$$dF = \frac{1}{4\pi\epsilon_0}\frac{q.dQ}{x^2}$$
The numerator multiples the two charges, \(Q\) and \(dQ\); the denominator \(x\) is the distance between the two charges.
To find the total force, add up all the forces from each little \(dQ\)’s by integrating from the near end of the line \(a\), to the far end \(a+L\).
$$F = \int_{a}^{a+L}d\vec{F}=\int_{a}^{a+L}\frac{1}{4\pi\epsilon_0}\frac{q.dQ}{x^2}$$
This equation includes both \(x\) and \(dQ\) as variables. To get down to a single independent variable, eliminate \(dQ\) by replacing it with the expression \(\frac{Q}{L}dx\) from above,
$$F =\int_{a}^{a+L}\frac{1}{4\pi\epsilon_0}\frac{q.Q}{L.x^2}dx$$
Move everything that does not depend on \(x\) outside the integral.
$$F =\frac{1}{4\pi\epsilon_0}\frac{q.Q}{L}\int_{a}^{a+L}\frac{1}{x^2}dx$$
And solve the integral,
$$\large F = \frac{1}{4\pi\epsilon_0} \frac{qQ}{a(a+L)}$$
Some things to notice about the solution:
- The numerator is the product of the test charge and the total charge on the line, which makes sense.
- The denominator has the form , created by a combination of distance to the near end and far end of the line. The \(a(a+L)\)form of the denominator emerges from the particular geometry of this example.
- If the point charge \(q\)moves very far away from the line, \(L\) becomes insignificant compared to \(a\), and the denominator approaches \(a^2\). So at a great distance, the line starts to resemble a far-off point charge, and as one would hope, the equation approaches Coulomb’s Law for two-point charges.
We’ll do a few more electrostatics problems with simple charge geometries. After that, the math gets really involved, so the common strategy with complex geometries becomes: break down the geometry into simpler versions we already know how to do, then merge the answers.
Strategies for applying Coulomb’s Law
Coulomb’s Law is a good choice for situations with point charges and/or simple symmetric geometries like lines or spheres of charge.
Since Coulomb’s Law is based on pairwise forces between charges, when faced with multiple (more than two) point charges,
- Work out the forces between each pair of charges.
- Finish with a vector addition to merge the pairwise forces into a single resultant force.
For a situation with distributed charge, creatively model the distributed charge as a collection of point charges,
- Invent a little \(dQ\)representing an infinitesimal charge within the region of distributed charge.
- Work out the forces pairwise between the point charge and each little \(dQ\).
- Sum up the forces with an integral. This is a vector sum to get the resultant force.
Electric Force Definition:
The electric force, also known as the electrostatic force, is the attraction or repulsion between charged particles due to their electric charges. It is one of the fundamental forces in nature, alongside gravity and the strong and weak nuclear forces. Electric force is responsible for the interactions between electrons and protons, as well as other charged particles.
Electric Force Equation:
The electric force between two charged particles can be calculated using Coulomb’s law. The equation is given by:
F = k * (|q1 * q2|) / r^2
Here, F represents the magnitude of the electric force, k is the electrostatic constant (9 x 10^9 Nm^2/C^2), q1 and q2 are the charges of the particles, and r is the distance between them.
Electric Force Formula:
In its simplest form, the electric force formula can be written as:
F = k * q1 * q2 / r^2
This formula expresses the force between two point charges. The sign of the force depends on the charges of the particles: like charges (same sign) repel each other, while opposite charges (different signs) attract.
Electric Force Example:
A common example of electric force is the interaction between an electron and a proton in an atom. The electron, negatively charged, is attracted to the proton, positively charged, forming an electric force that keeps the atom together. This force is responsible for the stability of matter.
Electric Force Calculator:
To simplify calculations involving electric force, you can use online electric force calculators. These tools allow you to input the values of the charges and distance, and they will provide you with the resulting electric force. This is especially helpful when dealing with complex or multiple charges.
Electric Force Unit:
The SI unit of electric force is the Newton (N). However, in some cases, the unit of force can be expressed in other forms such as dynes or pound-force, depending on the context. It’s important to use consistent units in calculations to ensure accurate results.
Electric Force in a Sentence:
In a sentence, electric force can be described as the attraction or repulsion between charged particles due to their electric charges. It is responsible for various phenomena, such as the interaction between charged objects and the functioning of electrical devices.
Electrical Force Symbol:
The symbol used to represent electric force is “F.” In equations or formulas, this symbol is used to denote the magnitude of the force.
Electric Force Facts:
– Electric force is much stronger than gravitational force. For example, the electric force between two protons is about 10^36 times stronger than the gravitational force between them.
– Electric force obeys the inverse square law, meaning that it decreases as the distance between charged particles increases.
– Electric force is responsible for phenomena like static electricity, lightning, and the functioning of electronic devices.
Application of Electric Force:
The applications of electric force are vast and crucial in various fields. Some notable applications include:
– Electric motors and generators: Electric force is harnessed to produce mechanical energy, powering machines and devices.
– Electrostatic precipitators: These devices use electric force to remove particulate matter from industrial emissions.
– Capacitors and batteries: Electric force is used to store and release electrical energy in these devices.
– Particle accelerators: Electric force is utilized to accelerate charged particles to high speeds for scientific research.
FAQs
What is the electric force formula?
What is the formula of electric force? Electric force formula can be obtained from Coulomb’s law as follows: $$\vec F = K\frac{q_0.q_1}{r^2}\hat{r}$$ Where \(\vec{F}\) is the electric force-directed between two charged bodies.
What are the examples of electric force?
Electric circuits, A charged bulb, Exertion of static friction which is formed when a cloth is being rubbed by a dryer.
What is the law of electric force?
Coulomb’s law states that the electrical force between two charged objects is directly proportional to the product of the quantity of charge on the objects and inversely proportional to the square of the separation distance between the two objects.
What is the symbol for electric force?
\(F_E\)
What are two applications of electric force?
- The Van de Graaff Generator.
- Xerography.
- Laser Printers.
- Ink Jet Printers and Electrostatic Painting.
- Smoke Precipitators and Electrostatic Air Cleaning.
Why is electric force important?
In general, electrostatic forces become important when particle material is electrically insulating so the electric charge can be retained. In electrophotography, electrostatic forces are utilized to move charged toner particles from one surface to another for the purpose of producing high-quality prints.
Calculate the magnitude of the electric force upon an electron is placed at point B?
To calculate the magnitude of the electric force on an electron placed at point B, you need to know the charges and distances involved in the situation. The electric force is given by Coulomb’s law, which states that the force is directly proportional to the product of the charges and inversely proportional to the square of the distance between them. If you have the necessary information, you can plug the values into the electric force equation F = k * (|q1 * q2|) / r^2, where F represents the force, k is the electrostatic constant, q1 and q2 are the charges, and r is the distance. By substituting the values and performing the calculation, you can determine the magnitude of the electric force.
How much work W is done by the electric force on the moving point charge?
To determine the work done by the electric force on a moving point charge, you need to know the force acting on the charge and the displacement it undergoes. The work done is given by the formula W = F * d * cos(theta), where W represents the work, F is the force, d is the displacement, and theta is the angle between the force vector and the displacement vector. By multiplying the magnitude of the force by the displacement and the cosine of the angle, you can calculate the work done by the electric force.
How does an electric current produce a magnetic force?
An electric current produces a magnetic force through the interaction between moving charges and magnetic fields. When electric charges flow through a conductor, such as a wire, they create a magnetic field around the conductor. The magnetic field lines form concentric circles around the wire. The interaction between the moving charges and the magnetic field results in a force known as the magnetic force. This force follows the right-hand rule, where the direction of the force is perpendicular to both the direction of the current and the magnetic field. The magnitude of the force depends on the strength of the current and the strength of the magnetic field.
What is the electric force formula?
The electric force formula is given by Coulomb’s law, which states that the force between two charged objects is directly proportional to the product of their charges and inversely proportional to the square of the distance between them. The formula is expressed as F = k * (|q1 * q2|) / r^2, where F represents the force, k is the electrostatic constant (9 x 10^9 Nm^2/C^2), q1 and q2 are the charges of the objects, and r is the distance between them.
Which factors affect the strength of the electric force between two objects?
The strength of the electric force between two objects depends on several factors:
– Magnitude of the charges: The greater the magnitude of the charges, the stronger the electric force.
– Distance between the objects: The electric force decreases as the distance between the objects increases. It follows an inverse square relationship.
– Permittivity of the medium: The electric force is also influenced by the properties of the medium between the objects, known as the permittivity. Different materials have different permittivities, affecting the electric force.
What is electric force with an example?
Electric force can be demonstrated through various examples. One example is the interaction between a positively charged balloon and a negatively charged object, such as hair. When the balloon is rubbed against hair, it becomes negatively charged. The negatively charged balloon and the positively charged hair experience an electric force of attraction, causing the hair to be pulled towards the balloon. This phenomenon showcases the electric force in action.
What is the magnitude of the electric force on the ball?
To determine the magnitude of the electric force on a ball, you need to know the charges involved and the distance between them. Using Coulomb’s law and the formula F = k * (|q1 * q2|) / r^2, where F represents the force, k is the electrostatic constant, q1 and q2 are the charges, and r is the distance, you can calculate the magnitude of the electric force on the ball.
What is the magnitude of the electric force on charge A?
To calculate the magnitude of the electric force on charge A, you need to consider the charges and distances involved. Using Coulomb’s law and the formula F = k * (|q1 * q2|) / r^2, where F represents the force, k is the electrostatic constant, q1 and q2 are the charges, and r is the distance, you can determine the magnitude of the electric force on charge A.
What is called electric force?
The electric force, also known as the electrostatic force, is the attraction or repulsion between charged objects due to their electric charges. It is one of the fundamental forces in nature and plays a crucial role in various phenomena, such as static electricity, the behavior of atoms, and the functioning of electrical devices.
