Table of Contents
Free Equilibrium Constant Calculator provides a free online tool for displaying equilibrium constants for given chemical reactions. STUDYQUERIES’s online equilibrium constant calculator tool speeds up the calculation and displays the result in a fraction of a second.
How to Use the Equilibrium Constant Calculator?
To use the equilibrium constant calculator, follow these steps:
- Step 1: Enter the reactants, products, and their concentrations in the input fields
- Step 2: Click “Calculate Equilibrium Constant” to get the results
- Step 3: The equilibrium constant for the given chemical reaction will be displayed in the output field
Equilibrium Constant Calculator
What Is Equilibrium Constant And Calculator?
Homogeneous Reactions
Homogeneous reactions occur when the states of matter of the products and reactions are the same (homo means “same”). Solvents determine the state of matter for the reaction in most cases. By way of example, the synthesis of methanol from carbon monoxide and hydrogen is a gaseous homogeneous mixture, which contains two or more substances:
$$CO_{(g)}+2H_2{_{(g)}}\rightleftharpoons CH_3OH_{(g)}$$
The arrows show that at equilibrium, the forward and reverse reactions occur at equal rates. As the reaction reaches equilibrium, the equilibrium constant gives the ratio of the units (pressure or concentration) of the products to the reactants.
Another example of a gaseous homogeneous mixture is the synthesis of ammonia:
$$N_2{_{(g)}}+3H_2{_{(g)}}\rightleftharpoons 2NH_3{_{(g)}}$$
Heterogeneous Reactions
In a heterogeneous reaction, one or more states of the reaction differ (the Greek word “heteros” means “different”). Creating an aqueous solution of lead(II) iodide for example creates a heterogeneous mixture of particles in both the solid and aqueous states:
$$PbI_2{_{(s)}}\rightleftharpoons Pb^{+2}{_{(aq)}}+2I^{-3}{_{(aq)}}$$
Another example of a heterogeneous mixture is the decomposition of sodium hydrogen carbonate (baking soda) at high elevations. This reaction involves molecules in both solid and gaseous state:
$$2NaHCO_3{_{(s)}}\rightleftharpoons Na_2CO_3{_{(s)}}+H_2O{_{(g)}}+CO_2{_{(g)}}$$
$$C{_{(s)}}+O_2{_{(g)}}\rightleftharpoons CO_2{_{(g)}}$$
When approximating the activities of substances in equilibrium constant expressions, this difference between homogeneous and heterogeneous reactions is emphasized so that students remember that solids, pure liquids, and solvents are treated differently than gases and solutes.
You can reverse a reversible reaction both forwards and backward. In a closed system, most reactions are theoretically reversible, though some may be irreversible if they heavily favor the formation of products or reactants. The double half-arrow sign we use when writing reversible reaction equations,\(\rightleftharpoons\), is a good visual reminder that these reactions can go either forward to create products, or backward to create reactants. One example of a reversible reaction is the formation of nitrogen dioxide, \(NO_2\), from dinitrogen tetroxide, \(N_2O_4\):
$$N_2O_4(g)\rightleftharpoons NO_2(g)$$
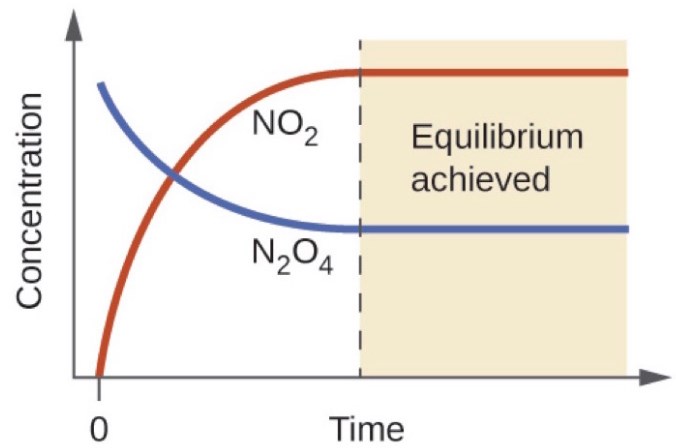
Imagine we added some colorless \(N_2O_4\) to an evacuated glass container at room temperature. If we kept our eye on the vial over time, we would observe the gas in the ampoule changing to a yellowish-orange color and gradually getting darker until the color stayed constant. We can graph the concentration of \(NO_2\) and \(N_2O_4\) over-time for this process, as you can see in the graph below.
Initially, the vial contains only \(N_2O_4\), and the concentration of \(NO_2\) is \(0\ M\). As \(N_2O_4\) gets converted to \(NO_2\) increases up to a certain point, indicated by a dotted line in the graph to the left, and then stays constant. Similarly, the concentration of \(N_2O_4\) decreases from the initial concentration until it reaches the equilibrium concentration. When the concentrations of \(NO_2\) and \(N_2O_4\) remain constant, the reaction has reached equilibrium.
Chemical equilibrium is reached when both forward and reverse reactions are taking place at the same rate. At equilibrium, the concentrations of the reactants and products are constant since the forward and reverse rates are equal. Remember that even though the concentrations at equilibrium are constant, the reaction still occurs! This is why this state is sometimes called dynamic equilibrium.
Based on the concentrations of all the different reaction species at equilibrium, we can define a quantity called the equilibrium constant \(K_c\), which is also sometimes written as \(K_{eq}\) or \(K\). The \(c\) in the subscript stands for concentration since the equilibrium constant describes the molar concentrations, in \(\frac{mol}{L}\), at equilibrium for a specific temperature. The equilibrium constant can help us understand whether the reaction tends to have a higher concentration of products or reactants at equilibrium. We can also use \(K_c\) to determine if the reaction is already at equilibrium.
How Do We Calculate Kc?
Consider the balanced reversible reaction below:
$$aA+bB\rightleftharpoons cC+dD$$
If we know the molar concentrations for each reaction species, we can find the value for \(K_c\) using the relationship
$$K_c=\frac{\left[C \right]^c\left[D \right]^d}{\left[A \right]^a\left[B \right]^b}$$
where \(\left[C \right]\) and \(\left[D \right]\) are equilibrium product concentrations; \(\left[A \right]\) and \(\left[B \right]\) are equilibrium reactant concentrations;\(a\), \(b\), \(c\) and \(d\) are the stoichiometric coefficients from the balanced reaction. The concentrations are usually expressed in molarity, which has units of \(\frac{mol}{L}\).
The following factors should be considered when calculating \(K_c\):
- \(K_c\) is a constant for a specific reaction at a specific temperature. If you change the temperature of a reaction, then \(K_c\) also changes.
- The equilibrium expression excludes pure solids and pure liquids, including solvents.
- \(K_c\) is often written without units, depending on the textbook.
- The reaction must be balanced with the coefficients written as the lowest possible integer values in order to get the correct value for \(K_c\).
Note: If any of the reactants or products are gases, we can also express the equilibrium constant as a partial pressure. We typically refer to that value as \(K_p\) to tell it apart from the equilibrium constant using concentrations in molarity, \(K_c\).
What does the magnitude of \(K_c\) tell us about the reaction at equilibrium?
The magnitude of \(K_c\) can give us some information about the reactant and product concentrations at equilibrium:
- If \(K_c\) is very large, ~1000 or more, we will have mostly product species present at equilibrium.
- If \(K_c\) is very small, ~0.001 or less, we will have mostly reactant species present at equilibrium.
- If \(K_c\) is in between 0.001 and 1000, we will have a significant concentration of both reactant and product species present at equilibrium.
By using these guidelines, we can quickly estimate whether a reaction will strongly favor the forward direction to make products—very large \(K_c\) -strongly favor the backward direction to make reactants, very small \(K_c\) or somewhere in between.
How Do We Calculate Kp?
The amount of a chemical at equilibrium can also be expressed in terms of its partial pressure when the reaction component is a gas. A gas equilibrium constant expressed in terms of partial pressure is written as \(K_p\).
Let’s take the generic balanced gas-phase reaction below as an example:
$$aA_{(g)}+bB_{(g)}\rightleftharpoons cC_{(g)}+dD_{(g)}$$
In this equation, \(a\) moles of reactant \(A\) react with \(b\), moles of reactant \(B\) to make \(c\) moles of product \(C\) and \(d\) moles of product \(D\).
If we know the partial pressures for each component at equilibrium, where the partial pressure of \(A_{(g)}\) is abbreviated as \(P_A\), then the expression for \(K_p\) for this reaction is
$$K_p=\frac{\left[P_C \right]^c\left[P_D \right]^d}{\left[P_A \right]^a\left[P_B \right]^b}$$
Remember the following important points when calculating \(K_p\):
- Ensure that the reaction is balanced! If not, the stoichiometric coefficients and exponents in the equilibrium constant will be incorrect.
- Pure liquids or solids have a concentration of \(1\) in the equilibrium expression. This is the same as when calculating \(K_c\).
- \(K_p\) is often written without units. Since the value of \(K_p\) depends on the units used for the partial pressure, you will need to check the pressure units used in your textbook when solving a \(K_p\) problem.
- All the partial pressures used for calculating \(K_p\) should have the same units.
- We can write \(K_p\) for reactions that include solids and pure liquids since they do not appear in the equilibrium expression.
Conversion of Kc to Kp
We can convert between gas concentration—in units of \(M\) or \(\frac{mol}{L}\),—and partial pressure using the ideal gas equation. Since molar concentration is the number of moles of gas per volume, or \(\frac{n}{V}\), we can rearrange the ideal gas equation to get the relationship between \(P\) and \(\frac{n}{V}\) as follows:
$$PV=nRT$$
Divide both sides by V.
$$\frac{PV}{V}=\frac{nRT}{V}$$
$$P=\frac{n}{V}RT$$
We can use this relationship to derive an equation to convert directly between \(K_c\) and \(K_p\) at temperature \(T\), where \(R\) is the gas constant:
$$K_p=K_c {(RT)}^{\Delta n}$$
The symbol \(\Delta n\) is the number of moles of gas on the product side minus the number of moles of gas on the reactant side in the balanced reaction:
$$\Delta n=(mol\ of\ product\ gas) – (mol\ of\ reactant\ gas)$$
Reaction Quotient
Another quantity of interest is the reaction quotient, \(Q\), which is the numerical value of the ratio of products to reactants at any point in the reaction. The reaction quotient is calculated the same way as is \(K\) but is not necessarily equal to \(K\). By predicting the reaction at any given point in time, it is possible to predict which way the reaction will proceed.
$$Q=\frac{[G]^g[H]^h}{[A]^a[B]^b}$$
- If \(Q>K\), then the reactions shift to the left to reach equilibrium
- If \(Q<K\), then the reactions shift to the right to reach equilibrium
- If \(Q=K\) then the reaction is at equilibrium
The same process is employed whether calculating \(Qc\) or \(Qp\).
Equilibrium Constant Expression Calculator:
The equilibrium constant expression calculator is a tool used to calculate the numerical value of the equilibrium constant for a chemical reaction. It takes into account the concentrations or partial pressures of the reactants and products at equilibrium. The equilibrium constant expression is determined by the stoichiometry of the balanced chemical equation and is denoted by K.
Example:
Consider the following chemical reaction:
2A + 3B ⇌ 4C
The equilibrium constant expression for this reaction is:
K = [C]^4 / ([A]^2 [B]^3)
Using the equilibrium constant expression calculator, you can input the concentrations of A, B, and C at equilibrium and obtain the value of K.
Equilibrium Constant Equation Calculator:
The equilibrium constant equation calculator is a tool used to calculate the equilibrium constant for a chemical reaction using experimental data. It involves determining the ratio of the concentrations or partial pressures of the products to the reactants at equilibrium. This calculator uses the experimentally obtained data points to find the equilibrium constant.
Example:
Suppose you have experimental data for a reaction where the initial concentrations of reactants and the equilibrium concentrations of products are known. By using the equilibrium constant equation calculator, you can input the data and calculate the value of the equilibrium constant, K.
What Is Kc Formula?
The kc formula refers to the mathematical expression used to calculate the equilibrium constant (Kc) for a chemical reaction in terms of concentrations. The formula is derived from the law of mass action and is specific to reactions in the gas or aqueous phase.
Example:
For the generic chemical reaction:
aA + bB ⇌ cC + dD
The Kc formula is given as:
Kc = ([C]^c [D]^d) / ([A]^a [B]^b)
Here, [A], [B], [C], and [D] represent the concentrations of A, B, C, and D, respectively, at equilibrium.
Equilibrium Concentrations Calculator:
The equilibrium concentrations calculator is a tool used to determine the concentrations of reactants and products at equilibrium for a given chemical reaction. It involves solving the equilibrium expressions and using the initial concentrations and stoichiometry of the reaction.
Example:
Consider the reaction:
2A + B ⇌ 3C
By using the equilibrium concentrations calculator, you can input the initial concentrations of A and B, as well as the equilibrium constant (Kc), and obtain the equilibrium concentrations of A, B, and C.
Equilibrium Constant Calculator From Delta G:
The equilibrium constant calculator from Delta G is a tool used to calculate the equilibrium constant (K) for a chemical reaction using the change in Gibbs free energy (∆G) of the reaction. The relationship between K and ∆G is given by the equation: ∆G = -RT ln(K), where R is the gas constant and T is the temperature in Kelvin.
Example:
If you know the value of ∆G for a reaction and the temperature, you can use the equilibrium constant calculator from Delta G to determine the corresponding value of K.
Equilibrium Constant Calculator With Temperature:
The equilibrium constant calculator with temperature is a tool that takes into account the effect of temperature on the equilibrium constant (K) of a chemical reaction. The calculator allows you to calculate the equilibrium constant at a specific temperature or to determine how the equilibrium constant changes with temperature.
Example:
By inputting the equilibrium constant value at one temperature and the corresponding temperature, you can use the equilibrium constant calculator with temperature to predict the value of K at a different temperature using the Van’t Hoff equation.
Equilibrium Calculator Physics:
The equilibrium calculator in physics refers to a tool used to solve problems related to static equilibrium in physics.
It involves analyzing the forces and torques acting on an object to determine whether it is in equilibrium or not.
Example:
Suppose you have a physics problem involving a system in static equilibrium, such as a beam supported by multiple forces. The equilibrium calculator in physics allows you to input the forces and torques acting on the system and determine if it is in equilibrium or if there is an unbalanced force or torque.
Equilibrium Ice Table Calculator:
The equilibrium ice table calculator is a tool used in chemistry to determine the equilibrium concentrations of species in a chemical reaction using an ICE table (Initial, Change, Equilibrium table). The ICE table tracks the changes in concentrations of reactants and products as the reaction reaches equilibrium.
Example:
Consider the reaction:
2A + B ⇌ 3C
By using the equilibrium ice table calculator, you can input the initial concentrations of A and B and determine the equilibrium concentrations of A, B, and C by following the steps of constructing and solving an ICE table.
Balanced Equilibrium Equation Calculator:
The balanced equilibrium equation calculator is a tool used to balance a chemical equation representing a reaction at equilibrium. Balancing the equation is necessary to determine the stoichiometric coefficients and to write the equilibrium constant expression accurately.
Example:
Suppose you have an unbalanced chemical equation representing an equilibrium reaction. By using the balanced equilibrium equation calculator, you can input the unbalanced equation, and the calculator will balance it by adjusting the coefficients to ensure that the number of atoms on both sides of the equation is equal.
FAQs
What is the equilibrium constant K?
The equilibrium concentrations of all reactants and products can be measured in a reaction at equilibrium. Equation K shows how the concentrations of the products depend on the concentrations of the reactants.
What is an equilibrium constant simple definition?
Calculates the relationship between the amounts of products and reactants present at equilibrium in a reversible chemical reaction at a given temperature.
Is equilibrium constant Kp or Kc?
The equilibrium constant Kc is given as a ratio between the concentrations of products and reactants. The equilibrium constant, Kp, is given as the ratio between the pressures of products and reactants.
What does K << 1 mean in chemistry?
If K>>1, the mixture will be mostly a product. If K<<1, the mixture will be mostly reactant. If K is about 1, the reaction will reach equilibrium at some intermediate mixture.
What are Kp and Kc?
The equilibrium constants of an ideal gaseous mixture are Kp and Kc. When equilibrium concentrations are expressed in atmospheric pressure, Kp is the equilibrium constant. When equilibrium concentrations are expressed in molarity, Kc is the equilibrium constant.
How To Find Equilibrium Constant With Graphing Calculator?
To find the equilibrium constant with a graphing calculator, you would need to plot a graph using experimental data. The graph typically represents the concentration or partial pressure of the products versus the concentration or partial pressure of the reactants at different points in time. The equilibrium constant can be determined by analyzing the slope or intercept of the graph.
How Do I Calculate The Equilibrium Constant?
To calculate the equilibrium constant, you need the concentrations (or partial pressures) of the reactants and products at equilibrium. The equilibrium constant expression is derived from the balanced chemical equation. Once you have the concentrations, you substitute these values into the equilibrium constant expression to obtain the numerical value of the equilibrium constant.
How To Calculate Equilibrium Constant Kp?
To calculate the equilibrium constant (Kp), you need the partial pressures of the reactants and products at equilibrium. The equilibrium constant expression for Kp is similar to that of Kc but uses partial pressures instead of concentrations. You substitute the partial pressure values into the equilibrium constant expression to calculate the numerical value of Kp.
Calculating Equilibrium Composition When Constant Is Known Calculator?
The calculating equilibrium composition when the constant is known calculator helps determine the concentrations of reactants and products at equilibrium when the equilibrium constant (K) is known. By using the known value of K and the stoichiometry of the balanced chemical equation, the calculator enables you to calculate the equilibrium composition.
Write The Equilibrium Constant Expression For This Reaction Calculator?
The “Write the equilibrium constant expression for this reaction” calculator is a tool that assists in determining the correct equilibrium constant expression for a given chemical reaction. It requires inputting the balanced chemical equation, and the calculator will generate the corresponding equilibrium constant expression based on the stoichiometry of the reaction.
How To Calculate Gibbs Free Energy With Equilibrium Constant Calculator?
The Gibbs free energy (∆G) can be calculated from the equilibrium constant (K) using the equation ∆G = -RT ln(K), where R is the gas constant and T is the temperature in Kelvin. The “calculate Gibbs free energy with equilibrium constant calculator” allows you to input the equilibrium constant value and the temperature to calculate the corresponding ∆G value.
How To Calculate Equilibrium Constant On A Calculator?
To calculate the equilibrium constant on a regular calculator, you need the concentrations (or partial pressures) of the reactants and products at equilibrium. Then, substitute these values into the equilibrium constant expression and perform the necessary mathematical operations (such as division or exponentiation) to calculate the numerical value of the equilibrium constant manually.

