Table of Contents
An online tool called pH Calculator displays a chemical solution’s pH value when entered. With StudyQueries’s pH calculator, you can calculate pH quickly, and the measurement appears in seconds.
How to Use pH Calculator?
To use the pH calculator, follow these steps:
- Step 1: Enter the name of the chemical solution and its concentration value in the respective input fields
- Step 2: Click on the “Calculate” button to obtain the pH value
- Step 3: Finally, the pH value will be visible in a new window
pH Calculator
What is Meant by pH Measurement?
Hydrogen potential is represented by the term pH in chemistry. Acidity and alkalinity are measured by pH. This is a measure of hydrogen ion concentration, which determines the acidity or alkalinity of a chemical solution. There is usually a pH range of 0 to 14 on the pH scale.

pH values greater than 7 are considered acidic for an aqueous solution at 25°C, while pH values less than 7 are considered alkaline or base for the solution.
Consider the chemical acetic acid, which has a molecular weight of 5. Therefore, the pH value of 5M acetic acid is 2.03.
Acids and bases
pH is a measurement of the hydrogen ion concentration in a solution relative to pure water. This determines whether a solution is acidic or basic. Acidic solutions have a higher H+ concentration than water (greater than 1 × 10^{-7} M), while basic (alkaline) solutions have a lower H+ concentration (less than 1 × 10^{-7} M). Hydrogen ion concentrations in solutions are typically expressed using pH values. A pH value is calculated as the negative log of hydrogen ion concentration:
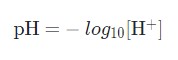
We are referring to its concentration by the square brackets around the H+. If you plug the hydrogen ion concentration of water (1 × 10^{-7} M) into this equation, you’ll get a value of 7.0, also known as neutral pH. In the human body, both blood and the cytosol (watery goo) inside of cells have pH values close to neutral. Aqueous (water-based) solutions gain and lose H+ concentration as acids or bases are added.
In our context, an acid is a substance that increases the concentration of hydrogen ions (H+) in a solution, usually by dissociating one of its hydrogen atoms. By contrast, a base raises pH by removing hydrogen ions from the solution using hydroxide (OH-) or another ion or molecule. (This simplified explanation of acids and bases is best used when thinking about biology. You might also want to look at the chemistry section for more information on acids and bases.)
The stronger the acid, the more readily it dissociates to form H+. If hydrochloric acid (HCl) is placed in water, it completely dissociates into hydrogen and chloride ions, so it is considered a strong acid. Tomato juice and vinegar, on the other hand, do not completely dissociate in water and are therefore considered weak acids. Similar to strong bases, sodium hydroxide (NaOH) dissociates completely in water, releasing hydroxide ions (or other types of basic ions) capable of absorbing H+.
The pH scale
The pH scale is used to measure acidity and basicity (alkalinity) in solutions. A change in pH value corresponds to a tenfold change in the concentration of H+ ions since the scale is based on pH values. It is often said that the pH scale is 0 to 14, and most solutions do fall within this range, although it is possible to get a pH below 0 or above 14. Anything below 7.0 is acidic, while anything above 7.0 is alkaline, or basic.
Both the pH inside human cells (6.8) and the pH of blood (7.4) are very close to neutral. It is usually considered unfavorable for life for pH values to exceed or to fall below the normal range. Although your stomach is highly acidic, with a pH of 1 to 2, the environment inside it is highly alkaline. What is the stomach’s solution to this problem? Cells that can be thrown away are the answer! Cells in the stomach, especially those that come in direct contact with stomach acid and food, are constantly dying and being replaced. Approximately every seven to ten days, the stomach lining is completely replaced.
Buffers
pH needs to remain within a fairly narrow range for most organisms, including humans. As an example, human blood must maintain a pH of 7.4 or lower throughout its lifetime and avoid a significant increase in acidity or alkalinity despite other ingredients entering or leaving the bloodstream.
Stable concentrations of H+ ions in biological systems are maintained by buffers, solutions that resist pH changes. As H+ ions accumulate in the water, a buffer absorbs some of them, bringing pH back to normal. Conversely, a buffer will donate some of its own H+ ions to bring pH down.
Buffers typically consist of an acid-base pair in which the acid and base differ by the presence or absence of a proton (a conjugate acid-base pair). Carbonic acid (H2CO3) and the bicarbonate ion (HCO3-) are important buffers that maintain the pH of human blood, for example.
In the blood, carbon dioxide is dissolved into carbonic acid when it combines with water in the bloodstream and moves from the muscles (where it is generated) to the lungs (where it is converted back into water and CO2, which is released as waste).
(H+)+ HCO3- > H2CO3
The equation above will shift right if there are too many H+ ions, and the bicarbonate ions will absorb the H+ ions to form carbonic acid. When the H+ concentration falls below a critical point, the equation has pulled the left and carbonic acid is converted to bicarbonate, which donates H+ to the solution. It would be impossible to survive if the pH of the body fluctuated enough to jeopardize survival.
Pool pH Calculator:
The pool pH calculator is a tool used to determine the pH level of a swimming pool. The pH level of a pool is important for maintaining water clarity, preventing corrosion of pool equipment, and ensuring the effectiveness of sanitizers. The calculator typically takes into account factors such as the pool’s total alkalinity, cyanuric acid levels, and the desired pH range.
Example:
Let’s say you have a swimming pool with a total alkalinity of 100 ppm and a cyanuric acid level of 30 ppm. Using a pool pH calculator, you input these values along with your desired pH range of 7.2-7.6. The calculator then determines that you need to add a specific amount of pH increaser or pH reducer to achieve the desired pH level.
Solution:
Based on the calculator’s recommendations, you can add the appropriate amount of pH increaser (such as sodium bicarbonate) or pH reducer (such as muriatic acid) to the pool water. It’s important to carefully follow the instructions provided by the calculator and the product labels to ensure safe and accurate adjustments.
pH Calculator from Molarity:
This type of pH calculator allows you to determine the pH of a solution based on its molarity. Molarity is a measure of the concentration of a solute in a solution, expressed as the number of moles of solute per liter of solution. pH, on the other hand, is a measure of the acidity or alkalinity of a solution.
Example:
Suppose you have a solution with a molarity of 0.1 M. Using a pH calculator that considers the dissociation constant of water (Kw), you can determine the pH of the solution. The pH of a neutral solution is 7, so the pH calculator calculates the concentration of H+ ions in the solution based on the molarity.
Solution:
Using the pH calculator, you can find that a 0.1 M solution has a pH of 1 (since pH is the negative logarithm of the H+ concentration). This means the solution is highly acidic.
pKa to pH Calculator:
The pKa to pH calculator is a tool used to convert the pKa value of an acid or base to its corresponding pH value. The pKa is a measure of the acidity or basicity of a compound and is related to the equilibrium constant of its dissociation in water.
Example:
Let’s consider a weak acid with a pKa value of 4. Using the pKa to pH calculator, you input the pKa value, and the calculator converts it to the corresponding pH value.
Solution:
Using the pKa to pH calculator, you find that a weak acid with a pKa of 4 has a pH of 4. This means the solution is moderately acidic.
pH Solution Calculator:
The pH solution calculator is a tool used to determine the pH of a solution by taking into account the concentrations of various ions or compounds present in the solution. It considers the dissociation constants (Ka) of acids or bases and the concentrations of their conjugate species.
Example:
Suppose you have a solution that contains a weak acid with a known concentration and its conjugate base with another known concentration. Using a pH solution calculator, you input the concentrations and the dissociation constants of the acid and its conjugate base. The calculator then calculates the pH of the solution based on these values.
Solution:
Using the pH solution calculator, you can find the pH of the solution based on the concentrations of the acid and its conjugate base and their dissociation constants. The calculator takes into account the equilibrium between the acid and its conjugate base and determines the pH accordingly.
pH Calculator from H+ Concentration:
The pH calculator from H+ concentration allows you to determine the pH of a solution directly from the concentration of hydrogen ions (H+). The pH is calculated using the negative logarithm of the H+ concentration.
Example:
Suppose you have a solution with an H+ concentration of 1 x 10^-3 M. Using the pH calculator from H+ concentration, you input this value, and the calculator calculates the corresponding pH.
Solution:
Using the pH calculator, you find that a solution with an H+ concentration of 1 x 10^-3 M has a pH of 3. This indicates that the solution is acidic.
pH Calculator of Two Solutions:
The pH calculator of two solutions helps determine the pH when two solutions are mixed together. This type of calculator takes into account the concentrations and pH values of the individual solutions and calculates the resulting pH of the mixture.
Example:
Consider two solutions: Solution A with a pH of 3 and Solution B with a pH of 9. Using the pH calculator of two solutions, you input the pH values and concentrations of the solutions. The calculator then calculates the pH of the resulting mixture.
Solution:
Using the pH calculator, you find that when Solutions A and B are mixed, the resulting pH of the mixture depends on the concentrations of the individual solutions. The calculator accounts for the volume and concentrations of each solution to calculate the pH of the final mixture.
How Much Acid to Lower pH Calculator:
The “How much acid to lower pH” calculator helps determine the amount of acid needed to lower the pH of a solution by a specific amount. This type of calculator takes into account the current pH, desired pH, and the acid’s properties.
Example:
Suppose you have a solution with a pH of 7, and you want to lower it to a pH of 5. Using the “How much acid to lower pH” calculator, you input the current pH, desired pH, and the properties of the acid being used. The calculator then determines the amount of acid needed to achieve the desired pH reduction.
Solution:
Using the calculator, you find that to lower the pH from 7 to 5, a specific amount of acid needs to be added. The calculator takes into account the acid’s strength, concentration, and the volume of the solution to calculate the required amount.
pH Calculator with Ka:
The pH calculator with Ka allows you to determine the pH of a solution based on the dissociation constant (Ka) of a weak acid or base. The Ka value represents the equilibrium constant of the dissociation reaction.
Example:
Consider a weak acid with a Ka value of 1 x 10^-5. Using the pH calculator with Ka, you input the Ka value, and the calculator calculates the corresponding pH.
Solution:
Using the pH calculator, you find that a weak acid with a Ka of 1 x 10^-5 has a pH of 4.3. This indicates that the solution is slightly acidic.
Concentration to pH Calculator:
The concentration to pH calculator is used to determine the pH of a solution based on the concentration of the solute. This type of calculator takes into account the dissociation of the solute and calculates the resulting pH.
Example:
Suppose you have a solution with a concentration of 0.01 M of a weak acid. Using the concentration to pH calculator, you input the concentration and the acid’s dissociation constant. The calculator then calculates the pH based on these values.
Solution:
Using the concentration to pH calculator, you find that a 0.01 M solution of the weak acid has a pH of 2.6. This indicates that the solution is acidic.
pH Calculator from pKa:
The pH calculator from pKa allows you to determine the pH of a solution based on the pKa
value of a weak acid or base. The pKa is the negative logarithm of the acid dissociation constant (Ka).
Example:
Consider a weak acid with a pKa value of 4.5. Using the pH calculator from pKa, you input the pKa value, and the calculator calculates the corresponding pH.
Solution:
Using the pH calculator, you find that a weak acid with a pKa of 4.5 has a pH of 4.5. This indicates that the solution is moderately acidic.
pH Calculator Titration:
The pH calculator for titration is used to determine the pH during an acid-base titration process. It takes into account the volumes and concentrations of the acid and base solutions used, as well as the dissociation constants (Ka) of the involved acids or bases.
Example:
Suppose you have a titration setup where you are adding a strong base to a solution containing a weak acid with a known concentration and volume. Using the pH calculator for titration, you input the volumes and concentrations of the acid and base solutions, as well as their dissociation constants. The calculator then determines the pH at different points during the titration process.
Solution:
Using the pH calculator for titration, you can track the pH changes as the titration progresses. The calculator considers the stoichiometry of the acid-base reaction and determines the pH at various volumes of the added base, allowing you to understand the pH evolution during the titration process.
Frequently Asked Questions About pH Calculator
How do you solve for pH on a calculator?
The procedure to use the pH calculator is as follows:
Step 1: Enter the chemical solution name and its concentration value in the respective input field.
Step 2: Now click the button “Calculate” to get the pH value.
Step 3: Finally, the pH value will be displayed in the new window.
What are pH and its formula?
The pH and its negative logarithm are Sorensen’s definitions of hydrogen concentration and its pH. The pH of an aqueous solution can be calculated based on the hydronium ion concentration using the equation pH=−log[H3O+].
How do you calculate H+ from pH?
In the following equation, pH = −log [H+], where [H+] denotes the molar hydrogen ion concentration. In order to calculate pH, we must take the common (base 10) logarithm of hydrogen ion concentration.
What is a pH of 10?
The pH scale ranges from 0 to 14. A pH of 7 is neutral. A pH less than 7 is acidic. A pH greater than 7 is basic.
How Much Muriatic Acid to Lower pH Calculator?
To determine the amount of muriatic acid needed to lower the pH of a solution, you can use the following steps:
1. Determine the current pH of the solution.
2. Determine the desired pH you want to achieve.
3. Calculate the pH difference by subtracting the desired pH from the current pH.
4. Based on the strength and concentration of muriatic acid you have, consult a table or reference source to find the acid’s neutralizing power or acidification potential (usually given in terms of normality or molarity).
5. Use the neutralizing power value to calculate the amount of muriatic acid needed. Multiply the neutralizing power by the pH difference obtained in step 3.
6. Adjust the volume of the solution to be treated. If you are working with a large body of water, such as a pool, refer to recommended guidelines for acid addition and dilution.
It’s important to note that muriatic acid is highly corrosive and should be handled with extreme caution. Always follow safety guidelines and wear appropriate protective equipment when working with acids.
How Much Acid to Lower pH Calculator?
The calculation to determine the amount of acid needed to lower the pH of a solution depends on several factors, including the strength of the acid, the initial pH, the desired pH, and the volume of the solution.
Here’s a general approach to calculate how much acid is needed:
1. Determine the current pH of the solution.
2. Determine the desired pH you want to achieve.
3. Calculate the pH difference by subtracting the desired pH from the current pH.
4. Based on the acid’s strength and concentration, consult a reference source to find the acid’s acidification potential or neutralizing power (usually given in terms of normality or molarity).
5. Use the acidification potential to calculate the amount of acid needed. Multiply the acidification potential by the pH difference obtained in step 3.
6. Adjust the volume of the solution accordingly.
Keep in mind that different acids have different acidification potentials, so the calculations may vary depending on the specific acid you are using.
How Much Soda Ash to Raise pH Calculator?
To determine the amount of soda ash (sodium carbonate) needed to raise the pH of a solution, follow these steps:
1. Determine the current pH of the solution.
2. Determine the desired pH you want to achieve.
3. Calculate the pH difference by subtracting the current pH from the desired pH.
4. Based on the chemical properties of soda ash and its ability to raise pH, consult a reference source to find the recommended dosage or the alkalinity increase per unit of soda ash added.
5. Use the recommended dosage to calculate the amount of soda ash needed. Multiply the alkalinity increase per unit by the pH difference obtained in step 3.
6. Adjust the volume of the solution to be treated.
It’s important to note that soda ash can be caustic and should be handled with care. Follow safety guidelines and wear protective equipment when working with alkaline substances.
How Much Caustic to Raise pH Calculator?
To determine the amount of caustic substance (such as sodium hydroxide or potassium hydroxide) needed to raise the pH of a solution, the following steps can be followed:
1. Determine the current pH of the solution.
2. Determine the desired pH you want to achieve.
3. Calculate the pH difference by subtracting the current pH from the desired pH.
4. Consult a reference source or chemical supplier to obtain the appropriate dosage or alkalinity increase per unit of caustic substance added.
5. Use the recommended dosage to calculate the amount of caustic substance needed. Multiply the alkalinity increase per unit by the pH difference obtained in step 3.
6. Adjust the volume of the solution to be treated.
Caustic substances are highly reactive and can be hazardous. It is crucial to handle them with extreme caution, following safety protocols and wearing appropriate protective gear.
How Do You Calculate a pH?
To calculate the pH of a solution, follow these steps:
1. Determine the concentration of hydrogen ions (H+) in the solution. This can be given directly or obtained by analyzing the dissociation of acids or bases in the solution.
2. Take the negative logarithm (base 10) of the H+ concentration. The formula for pH is: pH = -log[H+].
3. Round the pH value to the desired number of decimal places, typically two decimal places.
For example, if the H+ concentration is 1 x 10^-4 M, the calculation would be:
pH = -log(1 x 10^-4) = -(-4) = 4
Therefore, the pH of the solution would be 4.
When pH Is 9?
When the pH of a solution is 9, it indicates that the solution is alkaline or basic. In other words, it has a higher concentration of hydroxide ions (OH-) than hydrogen ions (H+). A pH of 9 is considered moderately basic.
What Is the pH of 0.0015 M HCl Solution?
Hydrochloric acid (HCl) is a strong acid that dissociates completely in water. Since the concentration of HCl is 0.0015 M, we can assume that all of it ionizes to produce hydrogen ions (H+).
The pH of a solution with a known H+ concentration can be calculated using the formula: pH = -log[H+].
In this case, the concentration of H+ in the solution is 0.0015 M. Therefore:
pH = -log(0.0015) = -(-2.82) = 2.82
Therefore, the pH of a 0.0015 M HCl solution is approximately 2.82, indicating that it is strongly acidic.
