Table of Contents
Polymers Of Carbohydrates is a major account within the field of glycoscience and covers the study and exploitation of polysaccharides that have a current or potential function in areas such as bioenergy, bioplastics, biomaterials, biorefining, chemistry, drug delivery, food, health, nanotechnology, packaging, paper, pharmaceuticals, medicine, oil recovery, textiles, tissue engineering and wood, and other conditions of glycoscience.
The appearance of the well-characterized carbohydrate polymer must be the major proportion of the work detailed, not a peripheral topic.
For all polysaccharides, including those achieved from a supplier, essential structural information which will disturb their behavior in the subsequent study should be given. Examples of such essential information include molecular weight, man urinates/guluronate ratio for alginates, degree of esterification for pectin, degree of deacetylation for chitosan.
What Are The Monomers And Polymers Of Carbohydrates?
The most generous biomolecules on earth are carbohydrates. From a chemical aspect, carbohydrates are principally a combination of carbon and water, and many of them have the empirical formula (CH2O)n, where n is the number of repeated groups. This view shows these molecules easily as “hydrated” carbon atom chains in which water molecules connect to each carbon atom, forcing to the term “carbohydrates.”
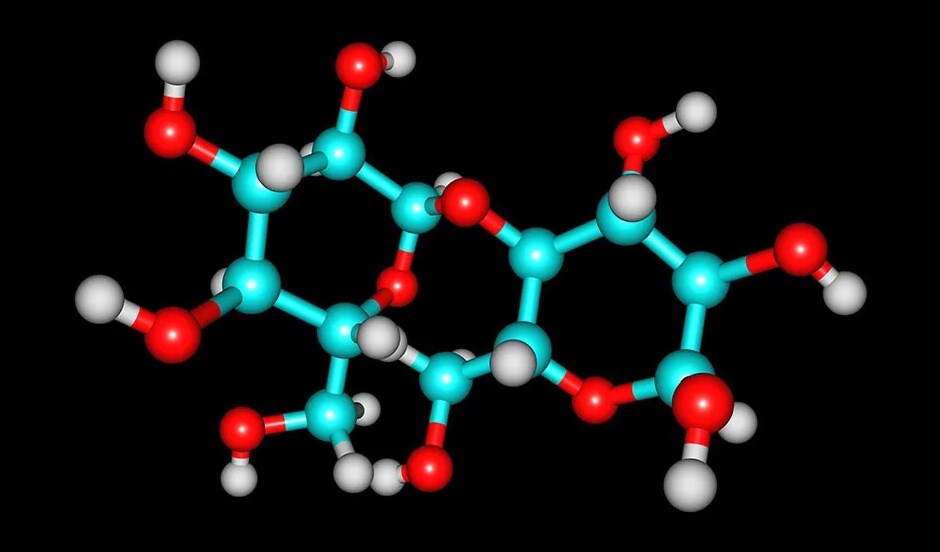
Although all carbohydrates contain carbon, hydrogen, and oxygen, there are some that also contain nitrogen, phosphorus, and/or sulfur. Carbohydrates have myriad contrasting functions. They are abundant in earthbound ecosystems, many patterns of which we use as food sources.
These molecules are also vital components of macromolecular structures that store and transmit genetic information (i.e., DNA and RNA). They are the basis of biological polymers that impart strength to various structural components of organisms (e.g., cellulose and chitin), and they are the elementary source of energy storage in the form of starch and glycogen.
In biochemistry, carbohydrates are generally called saccharides, from the Greek sakcharon, meaning sugar, although not all the saccharides are sweet. The most elementary carbohydrates are called monosaccharides, or simple sugars. They are the building blocks (monomers) for the synthesis of polymers or complex carbohydrates, as will be explained further in this post.
Types Of Polymers Of Carbohydrates
Monosaccharides are classified based on the number of carbons in the molecule. General categories are identified using a prefix that implies the number of carbons and the suffix –ose, which implies a saccharide; for example, triose (three carbons), tetrose (four carbons), pentose (five carbons), and hexose (six carbons). The hexose D-glucose is the most abundant monosaccharide in nature. Other very common and abundant hexose monosaccharides are galactose, used to make the disaccharide milk sugar lactose, and the fruit sugar fructose.
Monosaccharides
Monosaccharides are the monomers of carbohydrates and are often referred to as “the simple sugars”. There are dozens of them but they are classified conferring to the number of carbons they contain. For example, glucose has 6 carbons and belongs to a group of monosaccharides called the hexose sugars: “hex” meaning 6. Triose sugars, like glyceraldehyde, have 3 carbons.
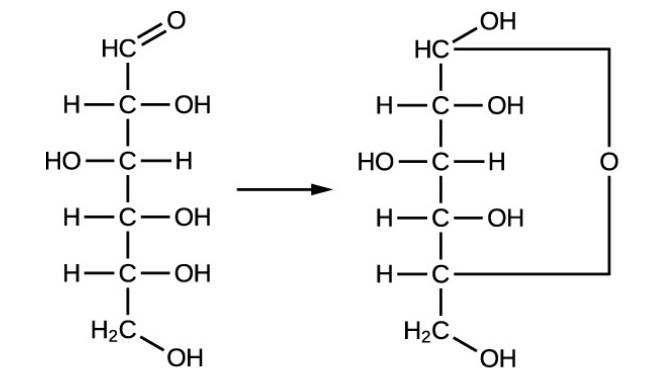
Pentose sugars, such as ribose, have 5. Monosaccharides range from 3 to 7 carbons. We are going to concentrate on 3 hexose sugars that organisms use for fuel: glucose, fructose, and galactose. Glucose, also recognized as dextrose, has the empirical formula C6H12O6. Glucose is the main energy source for life. We often call it blood sugar. Some organs in our body feed on nothing but glucose; our brain, for instance.
Fructose has the perfect same empirical formula as glucose: C6H12O6. Fructose is also known as levulose. Sweeter than the other 2 hexose sugars, fructose is commonly found in fruits and honey. Since fructose is sweeter, less is required, and so it is more economical.
High fructose corn syrup is now the considerable sugar source for soft drinks. Galactose, just like glucose and fructose, is C6H12O6. Galactose is rarely found free. Bonded to glucose, it forms lactose or milk sugar. Once ingested, both fructose and galactose are converted into glucose for an organism’s energy needs
Disaccharides
Two monosaccharide molecules may chemically bond to build a disaccharide. The name given to the covalent bond between the two monosaccharides is a glycosidic bond.
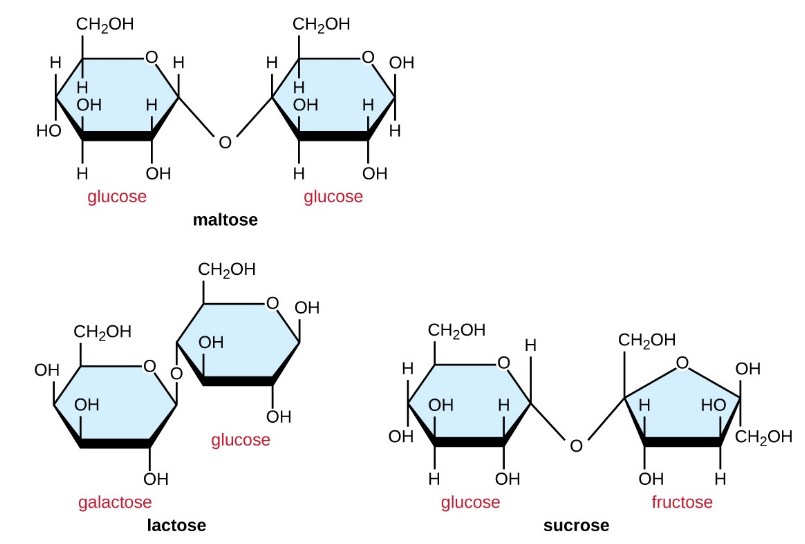
Glycosidic bonds form between hydroxyl groups of the two saccharide molecules, an example of the dehydration synthesis defined in the previous section of this post:
monosaccharide−OH+HO−monosaccharide⟶ (monosaccharide−O−monosaccharide) disaccharide
Common disaccharides are the grain sugar maltose, formed of two glucose molecules; the milk sugar lactose, formed of galactose and a glucose molecule; and the table sugar sucrose, formed of glucose and a fructose molecule.
Polysaccharides
Polysaccharides, also announced glycans, are large polymers composed of hundreds of monosaccharide monomers. Unlike mono- and disaccharides, polysaccharides are not sweet and, in familiar, are not soluble in water. Like disaccharides, the monomeric units of polysaccharides are linked together by glycosidic bonds.
Polysaccharides are very different in their structure. Three of the most biologically important polysaccharides—starch, glycogen, and cellulose—are all belonged to repetitive glucose units, although they differ in their structure. Cellulose subsists of a linear chain of glucose molecules and is a common structural factor of cell walls in plants and other organi*ms.
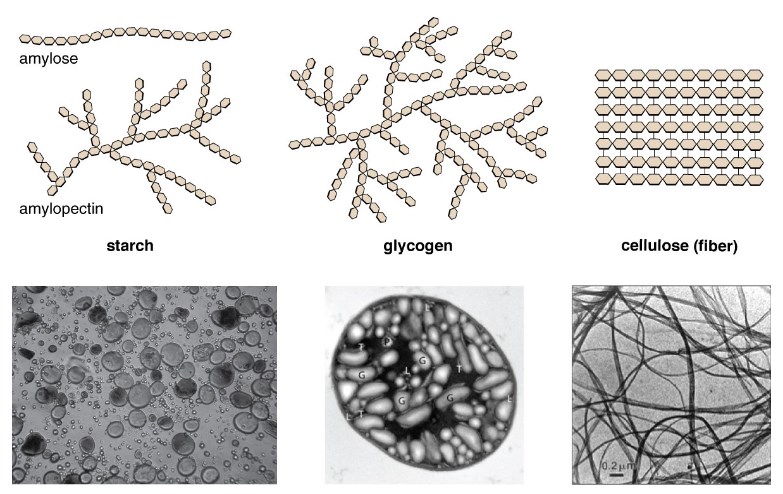
Glycogen and starch are branched polymers; glycogen is the primary energy-storage molecule in animals and bacteria, whereas plants primarily store energy in starch. The location of the glycosidic linkages in these three polymers is different as well and, as a consequence, linear and branched macromolecules have disparate properties.
Modified glucose molecules can be elemental components of other structural polysaccharides. Examples of these types of structural polysaccharides are N-acetyl glucosamine (NAG) and N-acetyl muramic acid (NAM) found in bacterial cell wall peptidoglycan. Polymers of NAG from chitin, which is found in fungal cell walls and in the exoskeleton of insects.
Starch: Starch is the reserved form of carbohydrate polymers in plants and is made up of a mixture of amylose and amylopectin (both polymers of glucose). Starch is made up of a monomer of carbohydrates – glucose that is joined by α 1-4 or α 1-6 glycosidic bonds.
The numbers 1-4 and 1-6 refer to the carbon number of the two residues that have joined to build the bond. Amylose is starch built by unbranched chains of glucose monomers (only α 1-4 linkages), whereas amylopectin is a branched polysaccharide (α 1-6 connections at the branch points).
Glycogen
Glycogen is the storage form of glucose in humans and other vertebrates and is made up of monomers of glucose.
Cellulose
Cellulose is the elementary structural polysaccharide in all plants and is a major unit in cell walls. It is a straight-chain polymer of β – ring structure of glucose that is held together by 1-4 glycosidic linkages.
Every other glucose monomer in cellulose is flicked over, and the monomers are packed tightly as continued long chains. This gives cellulose its rigidity and high tensile strength – which is so important to plant cells.
While the β 1-4 linkage cannot be cracked down by human digestive enzymes, herbivores such as cows, koalas, buffalos, and horses are able, with the help of the specialized flora in their stomach, to digest plant element that is rich in cellulose and use it as a food source.
What are the major functions of carbohydrates?
Carbohydrates supply abrupt and short-term energy. Carbohydrates are a basic material. Plants, fungi, bacteria, and arthropods build their bodies with it. Carbohydrates are the raw matter to make proteins, lipids, and nucleic acids. For example, plants make glucose first, and then all their other organic compounds are built from that.
Frequently Asked Question About Polymers Of Carbohydrates
