Table of Contents
Chemical Reaction Calculator displays a balanced equation, the structure, and all related data for any chemical reaction. STUDYQUERIES’s online chemical reaction calculator tool makes calculations faster and easier, displaying the equilibrium constant in a fraction of a second.
How to Use Chemical Reaction Calculator?
To use the chemical reaction calculator, follow these steps:
- Step 1: Enter the chemical reaction into the input field
- Step 2: Click “Submit” to receive the results
- Step 3: Finally, the balanced equation, structure, and all related data for the given chemical reaction will be displayed in a new window.
Chemical Reaction Calculator
What Is Chemical Reaction And Calculator?
Chemical reactions take place everywhere. Even though we sometimes associate chemical reactions with the sterile environment of the test tube and the laboratory, this couldn’t be further from the truth. In reality, the colossal number of transformations in our world every second makes for a dizzying array of new substances and energy changes.

In nature, chemical reactions are often less controlled and far messier than in the lab, and they often occur whether you want them to or not! Whether a forest fire burns, iron rusts in the presence of oxygen and water over a period of years, or fruit ripens on a tree, the process of turning one set of chemical substances (the reactants) into another set of chemicals (the products) is known as a chemical reaction.

Chemical reactions have been occurring on Earth since the dawn of time, but it wasn’t until the 18th century that the early chemists began to understand them. For centuries, fermentation, in which sugar is chemically converted into alcohol, has been known; however, its chemical basis has not been understood. How were these transformations controlled? When chemistry became a quantitative and experimental science, only then could these questions be answered.
Chemical Reaction Calculator: Historical Context Behind Chemical Equation
European and Persian philosophers began to be fascinated with the way that some substances were capable of “transforming” (or changing) into others in the early Middle Ages. The sulfur in simple stones burned magically, while otherwise unimpressive minerals, like the ore cinnabar, became a magical silvery liquid metal mercury when heated. Alchemists believed that there were four fundamental substances in the world: air, earth, fire, and water, based on Aristotle’s theories.
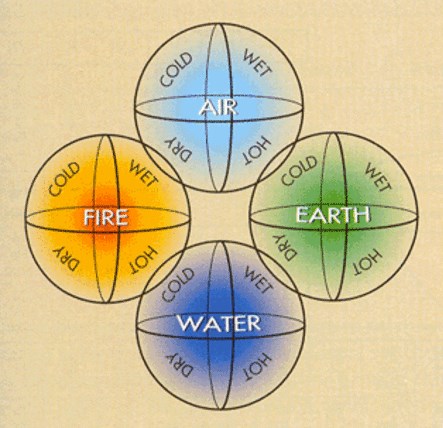
As a result, they proposed and spent generations trying to prove that copper and mercury, which were less expensive metals, could be turned into gold. Early alchemists performed chemical experiments – transforming one substance into another – despite their misguided approach, and it is hard to pinpoint a particular date or event that marked the arrival of the idea of a quantitative, orderly chemical reaction. In spite of this, there are some important moments in history that can help us understand it.
Law of Mass Conservation
Antoine Lavoisier was a French nobleman in the 1700s who began experimenting with chemical reactions. Chemists at the time couldn’t claim that chemistry was a quantitative science. The majority of theories that explained how substances changed rested on Greek philosophy, and there was little experimental detail associated with the alchemists’ experiments.
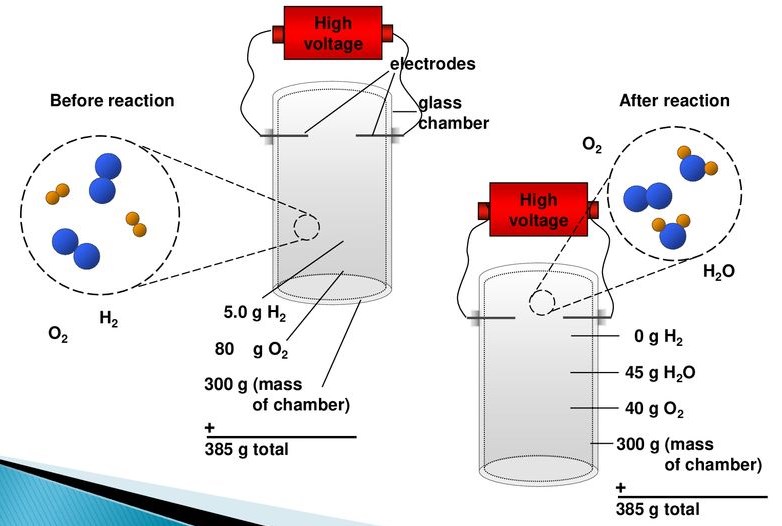
While Lavoisier conducted many quantitative experiments during the second half of the 18th century, he found that while substances changed form during chemical reactions, the mass of the system – or the total amount of “stuff” in the system – did not. Lavoisier was thus a proponent of the conservation of mass during transformations.
According to Lavoisier, unlike previous alchemists who thought they were creating matter from nothing, substances are neither created nor destroyed, but rather undergo chemical reactions during which they change form. It is widely regarded as the birth of modern chemistry as a quantitative science since Lavoisier published the seminal work Traité élémentaire de Chimie in 1789 (Lavoisier, 1789).
Law of Constant Composition
Joseph Proust was a French actor who followed Lavoisier’s footsteps. A number of chemical reactions were conducted by Proust, starting with different amounts of various substances. As he observed over time, no matter how he started a certain chemical reaction, the ratio in which the reactants were consumed remained constant.
For example, he worked extensively with copper carbonate, and copper, carbon, and oxygen all reacted together in a constant ratio regardless of how he changed the ratio of starting reactants (Proust, 1804). Proust, therefore, formulated the law of constant composition during the last few years of the 18th century.
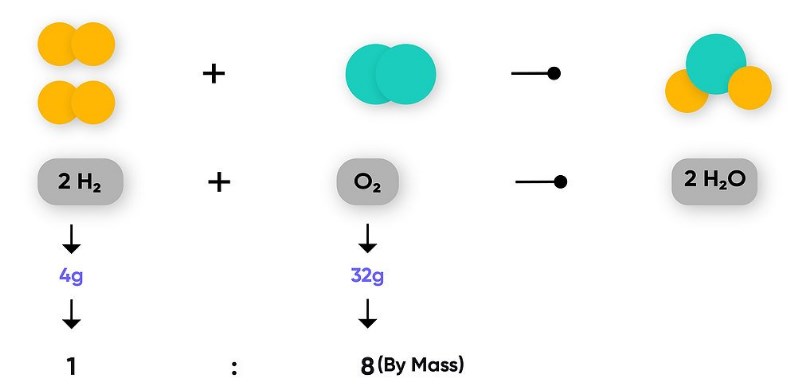
Regardless of the method of preparation, any given chemical substance (that we now define as a compound) always consisted of the same mass ratio of its elemental components. It was an important step forward in modern chemistry since it had previously been believed that the substances formed during chemical reactions were random and disordered.
Law of Multiple Proportions
English chemist John Dalton helped explain the laws of conservation of mass and definite ratios in 1803 by suggesting that matter is composed of atoms of unique substances that could not be created or destroyed (see our module Early Ideas about Matter for more information).
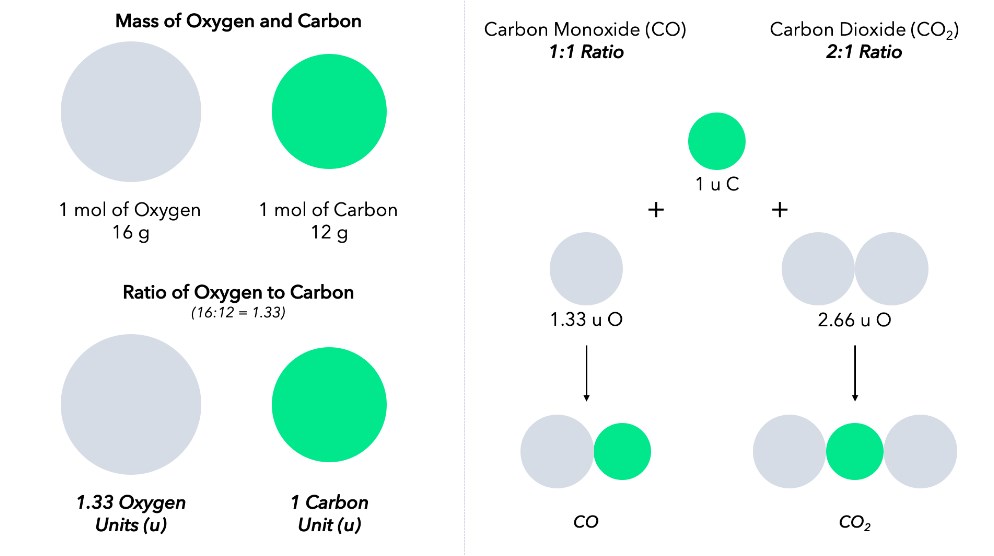
Proust’s ideas were extended by Dalton by recognizing that two elements could form more than one compound, but that no matter what the compound was, it would always have a whole-number ratio of elements (Dalton, 1808). With the help of Lavoisier’s atomic theory, this observation became known as the law of multiple proportions.
These advances laid the foundation for our modern understanding of chemical reactions, chemical equations, and chemical stoichiometry, or the process of expressing the relative quantities of reactants and products in a chemical reaction.
Chemical Reaction Calculator: Types of Chemical Reactions
The number of chemical reactions is staggering. Chemical reactions occur constantly in our bodies, in plants and animals, in the air that we breathe, in the lakes and oceans that we swim in, and even in the soil on which we grow crops and build our homes. In fact, there are so many chemical reactions occurring that it would be impossible, if not impossible, to comprehend them all.
In order to understand them, one method is to categorize chemical reactions into a few general types. Placing reactions together based on their similarities is not an ideal system, but it does help us identify patterns, which allows us to make predictions about reactions we haven’t yet learned about. This module examines and provides some context for several categories of reactions, specifically: synthesis, decomposition, single replacement, double replacement, REDOX (including combustion), and acid-base reactions.
Regardless of the type of reaction, one universal truth applies to all chemical reactions. A chemical reaction, i.e., a change in chemical composition, must result in the production of a new substance. A new substance is nearly always formed in conjunction with an energy change and is often accompanied by physical or observable changes.
Different types of physical change can occur, such as the formation of bubbles of gas, a solid precipitate, or a change in color. Chemists use these changes as clues that a chemical reaction is taking place and as a trigger for further research.
Synthesis Reactions
Lavoisier’s work led to a better understanding of how different gases are made up of different elements. As a result, different gases were commonly mischaracterized as air or as air with missing parts – for instance, “inflammable air” or “dephlogisticated air.” Lavoisier thought differently and was convinced that these were different substances.
In his experiments, he combined dephlogisticated air with inflammable air and a spark, and he discovered that the two combined to form water. Lavoisier renamed inflammable air “hydrogen” from the Greek hydro for “water” and genes for “creator.” In so doing, Lavoisier was identifying a synthesis reaction. In general, a synthesis reaction is one in which simpler substances combine to form another more complex one.
$$2H_2{_{(g)}} + O_2{_{(g)}} \longrightarrow 2H_2O{_{(l)}}$$
The reaction of hydrogen and oxygen (which Lavoisier also renamed dephlogisticated air) forming water can be summarized by the chemical equation shown below (for more information on chemical equations see the section titled Anatomy of a chemical equation). This represents a simple synthesis reaction.
Decomposition Reactions
In 1774, the scientist Joseph Priestley was fascinated by a mineral called cinnabar – a brick red mineral. He found that when he placed the mineral under sunlight magnified by a powerful magnifying glass, a gas was produced which he called “exalted” since a candle in the gas burned brightly (Priestley, 1775). A decomposition reaction led Priestley to discover oxygen without him realizing it.
As opposed to synthesis reactions, decomposition reactions involve the breaking down of a compound into simpler compounds or even elements. Priestley used heat to break down mercury (II) oxide (cinnabar) into its individual elements for his oxygen. He formulated the following equation to describe the reaction.
$$2HgO{_{(s)}} \longrightarrow 2Hg{_{(l)}} + O_2{_{(g)}}$$
Single Replacement Reactions
One of the first practical batteries was invented by the British chemist and meteorologist John Daniell in 1836. He used a very common single replacement reaction in his battery. Although his early cells were complex, with ungainly parts and complex constructs, the chemistry behind them was quite simple.
A single constituent can replace another already present in a chemical compound in some chemical reactions. In the Daniell cell, zinc can substitute for copper in a solution of copper sulfate, which exchanges electrons that are used in the battery cell. The reaction is summarized as follows:
$$Zn{_{(s)}} + CuSO_4{_{(aq)}} \longrightarrow ZnSO_4{_{(aq)}} + Cu{_{(s)}}$$
A metal displacement occurs when one metal replaces another metal, and many types of batteries rely on metal replacement reactions. In addition, there are several other types of single replacement reactions, such as when a metal replaces hydrogen from acid or from water or when a halogen replaces another halogen in a salt compound.
Combustion Reactions
The controlled use of fire was a crucial development for early civilization. While it’s difficult to pin down the exact time that humans first tamed the combustion reactions that produce fire, recent research suggests it may have occurred at least a million years ago in a South African cave (Berna et al. 2012).
Combustion is nothing more than the reaction between a fuel (wood, oil, gasoline, etc.) and oxygen. In order for combustion to occur, fuel and oxygen must be present. There is, however, often a requirement for activation energy, which can be provided by a spark or source of ignition energy. The fire triangle is composed of fuel, oxygen, and energy, and the absence of any one of them means combustion will not occur.
In the modern world, most fuels are hydrocarbons – substances that contain both hydrogen and carbon. Plants produce hydrocarbons when they grow, which makes them an excellent fuel source. Other hydrocarbons are produced when plants and animals decay over time (such as natural gas, oil, and other substances). Hydrogen and carbon within these fuels combine with oxygen to produce two very familiar compounds, water, and carbon dioxide. The following is a simple example of methane, or natural gas, being burned:
$$CH_4{_{(g)}} + 2O_2{_{(g)}} \longrightarrow CO_2{_{(g)}} + 2H_2O{_{(l)}}$$
Heat and light are also products of combustion, and it is these products that we use to cook our food or heat our homes.
Reduction-Oxidation Reactions
There are four types of reactions listed above, each of which is a subcategory of a single type of chemical reaction called a redox reaction. In a redox reaction, reduction and oxidation occur simultaneously, hence the name. The processes of oxidation and reduction can be defined in different ways, but whatever definition is used, the two processes are symbiotic, i.e., they must happen together.
According to one definition, oxidation is the loss of electrons by a species, and reduction is the gain of electrons by a species. It is clear that the two processes must occur simultaneously. When a chemical substance loses electrons (and thus is oxidized), it must have another chemical substance that it can give those electrons to.
During this process, the second substance (that which gains electrons) is said to be reduced. The original species can never lose its electrons without an electron acceptor, and so no oxidation can occur. When the electron acceptor is present, it is reduced and the redox combination process is complete.
For redox reactions of this type, there are two equations – one to show the loss of electrons (oxidation), and the other to show the gain of electrons (reduction). As an example, consider the Daniell cell above
$$\color{blue}{Oxidation: Zn \longrightarrow Zn^{2+}} + \color{red}{2e^-}$$
$$\color{blue}{Reduction: Cu^{2+}} + \color{red}{2e^-} \longrightarrow \color{blue}{Cu}$$
In the first reaction, electrons are lost by zinc, and the same electrons are accepted by copper ions in the second reaction. The reactions can be combined to cancel out the electrons on either side of the reactions, creating the overall redox reaction:
$$Zn + Cu^{2+} \longrightarrow Zn^{2+} + Cu$$
There are other definitions of oxidation and reduction, but in every case, the two halves of the redox reaction remain symbiotic – one loses and the other gains. The loss of one species cannot occur without the gain of another.
Double Displacement Reactions
When soap cannot easily lather in water, the water is considered ‘hard’. In addition to making it difficult to form a lather, hard water causes a host of other problems. The buildup of compounds in water pipes (known as ‘scale’) can obstruct the flow of water and cause problems in industrial processes. The textile industry relies heavily on water. In these situations, the quality of the water has a profound effect on the final product, so controlling the composition of the water is essential.
In hard water, magnesium or calcium ions are dissolved in salts, such as magnesium chloride or calcium chloride. In a double displacement reaction, soap (sodium stearate) reacts with either of those salts to form soap scum, a precipitate that is insoluble.
The double displacement reaction (also known as a double replacement reaction) occurs when two ionic substances come together and swap partners. Generally speaking:
$$AB + CD \longrightarrow AD + CB$$
Where A and C are cations (positively charged ions), and B and D are anions (negatively charged).
In the case of the reaction of soap with calcium chloride, the reaction is:
$$CaCl_2{_{(aq)}} + 2Na(C_{17}H_{35}COO){_{(aq)}} \longrightarrow 2NaCl{_{(aq)}} + Ca(C_{17}H_{35}COO)_2{_{(S)}}$$
Solid calcium stearate is what we call soap scum; it is formed when the soluble sodium stearate salt (soap) reacts with calcium chloride in a double replacement reaction.
Acid-Base Reactions
Acid-base reactions are all around us, and even inside of us. Every day we encounter acids and bases, from the classic baking soda volcano in elementary school to digestion.
When an atom of hydrogen loses its only electron, it forms a positive ion, H+. Hydrogen ions are the basis for all acids, and one definition of an acid is that it is a hydrogen ion donor. Compounds such as the citric acid in lemon juice, the ethanoic acid in vinegar, or a typical laboratory acid like hydrochloric acid, all give their hydrogen ions away in chemical reactions known as acid-base reactions.
Acids are chemical opposites of bases, and bases are hydrogen ions that accept hydrogen ions. An acid-base reaction occurs whenever an acid donates a hydrogen ion to a base, for example, when hydrochloric acid donates a hydrogen ion to sodium hydroxide:
$$HCl{_{(aq)}} + NaOH{_{(aq)}} \longrightarrow H_2O{_{(l)}} + NaCl{_{(aq)}}$$
A closer look at this reaction reveals that in water the HCl gives away an H+ as shown below:
$$HCl{_{(aq)}} + H_2O{_{(l)}} \longrightarrow H_3O^+{_{(aq)}} + Cl^-{_{(aq)}}$$
The resulting species, H3O+ (the hydronium ion), can, in turn, act as an acid when it comes into contact with any species that can accept a hydrogen ion, such as hydroxide ions from sodium hydroxide:
$$H_3O^+{_{(aq)}} + NaOH{_{(aq)}} \longrightarrow 2H_2O{_{(l)}} + Na^+{_{(aq)}}$$
Combining both equations gives us, Equation can be re-written to show the individual ions that are found in solution, thus:
$$H^+{_{(aq)}} + Cl^-{_{(aq)}} + Na^+{_{(aq)}} + OH^-{_{(aq)}} \longrightarrow H_2O{_{(l)}} + Na^+{_{(aq)}} + Cl^-{_{(aq)}}$$
Removing the spectator ions from the equation above, we get the net ionic equation:
$$H^+{_{(aq)}} + OH^-{_{(aq)}} \longrightarrow H_2O{_{(l)}}$$
Any chemical reaction that forms water from the reaction between an acid and base as in equation #9e is known as a neutralization reaction.
Anatomy Of A Chemical Equation
Because chemical equations are shorthand for describing chemical reactions, they are always associated with chemical reactions. Those facts alone make equations important, but equations also play a big role in describing the quantitative aspect of chemistry, which is called stoichiometry.
Chemical reactions all follow the same basic format. Starting substances, or reactants, are shown with their chemical formula to the left of an arrow, with multiple reactants separated by plus signs. For instance:
$$C + O_2 \longrightarrow$$
To the right hand of the arrow, one finds the chemical formulas of the new substance or substances (known as the products) that are produced by the chemical reaction. In this case, since carbon dioxide is the result of burning carbon in the presence of oxygen:
$$\color{blue}{[Reactants]\ C + O_2} \longrightarrow \color{red}{CO_2\ [Products]}$$
Considering that reactions can result in both physical and chemical changes, each substance is given its own state symbol, written as a subscript to the right of the formula, which describes the physical form of the reactants and products. Solids are abbreviated as (s) and liquids as (l) and gases as (g) and aqueous substances as (aq), i.e., those dissolved in water.
$$C{_{(s)}} + O_2{_{(g)}} \longrightarrow CO_2{_{(g)}}$$
Finally, in order to ensure that this representation complies with the law of conservation of mass, the equation might have to be balanced by adding numbers in front of each species that create equal numbers of atoms of each element on either side of the equation. The formation of carbon dioxide from carbon and oxygen does not require adding such numbers (called the stoichiometric coefficients), since one carbon atom and two oxygen atoms are present on each side of the equation.
Energy changes
Chemical reactions are often driven by energy exchanges in nature. As a result, reactions can be divided into two groups – those that release energy and those that absorb it.
Those reactions that release energy into the surrounding environment are exothermic. The combustion reaction is an obvious example since the energy released by the reaction is converted into light and heat.
Endothermic reactions, on the other hand, absorb energy from the environment. One may need to heat up the reaction or add another form of energy to the system before seeing the reaction progress.
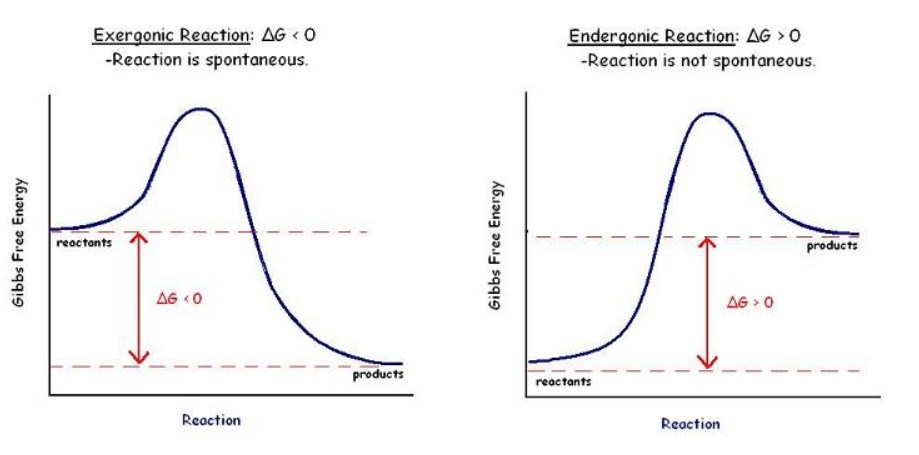
Energy is neither created nor destroyed in either case; rather, it is transferred from one type of energy to another, for instance, from chemical energy to heat or light. The energy that goes into the formation of chemical bonds is exchanged with the environment around that reaction for other forms of energy.
Photosynthesis is a classic example of this reaction, in which plants absorb light energy from the sun in order to create bonds between atoms that become sugars, which are then stored as chemical energy for later use. A plant uses the energy released from the breakdown of sugar molecules during respiration, which is the reverse of photosynthesis.
The Context Of Chemical Reactions
Every day, we are surrounded by chemical reactions. We experience chemical reactions in almost everything we do, whether it is a simple replacement reaction in a flashlight battery, a synthesis reaction when iron rusts with water and oxygen, or an acid-base reaction when we eat.
For a chemist working in a distant laboratory, understanding these reactions is not an abstract concept, but rather it is fundamental to understanding life. In order to truly master chemical reactions, we need to understand their quantitative nature, a concept known as stoichiometry, which we will discuss in another module.
Chemical Reaction Calculator: Important Concepts To Remember
- To better understand chemistry and chemical reactions, it was necessary to move from a qualitative to a quantitative approach.
- When a substance or substance (the reactants) undergoes a change that results in the formation of another substance or substance (the products), then a chemical reaction has occurred.
- Chemical reactions conserve mass and energy. It is neither created nor destroyed, rather it is conserved but rearranged to create new substances. Energy is neither created nor destroyed, instead, it is conserved and often converted into another form.
- Depending on their nature, chemical reactions can be classified into different types. As far as reactants and products are concerned, each type has its own distinctive characteristics.
- Chemical reactions often produce observable changes, such as energy changes, color changes, the release of gases, or the formation of solids.
- In chemical reactions, energy plays a crucial role. An exothermic reaction occurs when energy is released into the surroundings; an endothermic reaction occurs when energy is absorbed from the surroundings.
Chemical Reaction Product Calculator:
A chemical reaction product calculator is a tool used to predict the products of a chemical reaction based on the reactants entered. It takes the chemical equation as input and provides the corresponding products as output. For example, if you enter the reaction equation “2H2 + O2 → 2H2O,” the calculator will determine that the products are water (H2O).
Chemical Reaction Balance Calculator:
A chemical reaction balance calculator is used to balance chemical equations. It takes an unbalanced equation as input and adjusts the coefficients of the reactants and products to ensure that the law of conservation of mass is satisfied. For instance, if you enter the unbalanced equation “H2 + O2 → H2O,” the calculator will balance it to “2H2 + O2 → 2H2O.”
Chemical Reaction Equation Calculator:
A chemical reaction equation calculator is a tool that allows you to input the reactants and products of a chemical reaction and obtain the balanced chemical equation. For example, if you input the reactants as “Na + Cl2” and the products as “NaCl,” the calculator will provide the balanced equation “2Na + Cl2 → 2NaCl.”
Type of Chemical Reaction Calculator:
A type of chemical reaction calculator helps identify the type or category of a given chemical reaction. It analyzes the reactants and products to determine whether the reaction is a synthesis, decomposition, combustion, single replacement, double replacement, or other type of reaction. For example, if you input the reaction equation “2Na + Cl2 → 2NaCl,” the calculator will identify it as a synthesis reaction.
Chemical Reaction Type Identifier Calculator:
Similar to the previous calculator, a chemical reaction type identifier calculator is used to determine the specific type of chemical reaction based on the provided reactants and products. It helps classify reactions into more specific categories, such as acid-base, redox, precipitation, or gas-forming reactions. For instance, if you input the reactants as “HCl + NaOH” and the products as “NaCl + H2O,” the calculator will identify it as an acid-base reaction.
Find Product of Chemical Reaction Calculator:
A find product of chemical reaction calculator is used to determine the products of a given chemical reaction. It takes the reactants as input and calculates the resulting products. For example, if you enter the reactants as “Fe + O2,” the calculator will identify that the product is iron oxide (Fe2O3).
Chemical Reaction Identifier Calculator:
A chemical reaction identifier calculator is designed to recognize and classify different chemical reactions. It takes the reactants and products as input and provides information about the type of reaction, such as synthesis, decomposition, combustion, or displacement. For example, if you input the equation “CaCO3 → CaO + CO2,” the calculator will identify it as a decomposition reaction.
Organic Chemistry Reaction Solver:
An organic chemistry reaction solver is a specialized tool used to solve organic chemistry reactions. It helps predict the products of organic reactions based on the given reactants and reaction conditions. This type of calculator takes into account specific functional groups and reaction mechanisms to provide accurate results.
Predict Products of Chemical Reactions:
A predict products of chemical reactions calculator is used to anticipate the products that are likely to be formed in a chemical reaction. It takes the reactants as input and uses knowledge of chemical principles and reaction types to predict the resulting products. For example, if you input the reactants as “HCl + NaOH,” the calculator will predict that the products are NaCl and H2O.
Mass in Chemical Reaction Calculator:
A mass in chemical reaction calculator helps calculate the mass of reactants or products involved in a chemical reaction. It considers the stoichiometry of the balanced equation and allows you to input known masses or quantities to determine the unknown mass. For instance, if you know the mass of reactant A and want to find the mass of product B, the calculator will perform the necessary calculations based on the balanced equation.
Chemical Reaction Product Predictor:
A chemical reaction product predictor is a tool used to predict the products of a chemical reaction. It takes the reactants as input and applies chemical rules, principles, and knowledge of reaction types to generate the likely products. For example, if you enter the reactants as “C6H6 + O2,” the calculator will predict that the products are CO2 and H2O.
Word Equation to Chemical Equation Converter:
A word equation to chemical equation converter is used to convert a word equation, which describes a chemical reaction using words, into a balanced chemical equation. It takes the description of the reaction as input and provides the corresponding balanced equation as output. For example, if you input “sodium + water → sodium hydroxide + hydrogen,” the calculator will convert it to “2Na + 2H2O → 2NaOH + H2.”
Double Replacement Reaction Calculator:
A double replacement reaction calculator is specifically designed to handle double replacement reactions. It takes the reactants as input and determines the products based on the exchange of ions between the reactant compounds. For instance, if you input “AgNO3 + NaCl” as the reactants, the calculator will determine that the products are AgCl and NaNO3.
Chemical Reaction Calculator: Summary
The article explores the wide variety of chemical reactions by grouping them into general types. We review synthesis, decomposition, single replacement, double replacement, REDOX (including combustion), and acid-base reactions.
FAQs
What are the 4 types of chemical reactions?
Representation of four basic chemical reactions types: synthesis, decomposition, single replacement, and double replacement.
What are 5 examples of a chemical reaction?
Here are some examples of chemical changes:
- Burning wood
- Souring milk
- Mixing acid and base
- Digesting food
- Cooking an egg
- Heating sugar to form a caramel
- Baking a cake
- Rusting of iron
What is a chemical reaction give example?
One or more chemicals are changed into one or more other chemicals in a chemical reaction. For example, iron and oxygen combine to form rust. Baking soda and vinegar combine to make sodium acetate, carbon dioxide, and water.
What are simply chemical reactions?
A chemical reaction occurs when one or more substances, also known as reactants, are converted into one or more different substances, known as products. Chemical reactions differ from physical changes, such as ice melting into water and water evaporating into vapor.
What is a chemical reaction for kids?
When two different substances or elements are mixed together, a chemical reaction occurs. Afterward, they undergo a chemical reaction that turns them into a completely different substance.
Is breathing a chemical reaction?
The process of breathing involves the exchange of gases between an organism and its surroundings. The process of respiration is a chemical reaction between glucose and oxygen. Carbon dioxide and water are produced as waste products.
What is the Gypsum formula?
Gypsum is the name given to a mineral categorized as calcium sulfate mineral, and its chemical formula is calcium sulfate dihydrate, CaSO4⋅ 2H2O.
How To Find The Products Of A Chemical Reaction Calculator?
To find the products of a chemical reaction using a calculator, you would typically use a chemical equation calculator or a reaction product predictor. These calculators allow you to input the reactants of the reaction, and they will generate the corresponding products based on the reaction type and stoichiometry.
What Type Of Chemical Reaction Takes Place In A Calculator?
A calculator does not undergo a chemical reaction itself. It is an electronic device used for mathematical calculations. Therefore, the type of chemical reaction that takes place in a calculator is not applicable.
What Type Of Chemical Reaction Calculator?
A chemical reaction calculator is a tool that helps analyze and solve chemical reactions. It can determine the type of reaction, predict the products, balance the equation, convert between different forms (such as word equation to chemical equation), and perform other calculations related to chemical reactions. The type of chemical reaction calculator may vary depending on the specific features and capabilities of the calculator or software being used.
What Type Of Reaction Is Formed Chemical Calculator?
The type of reaction formed in a chemical calculator depends on the specific reaction being modeled or analyzed. A chemical calculator can handle various types of reactions, including synthesis, decomposition, combustion, displacement, acid-base, redox, and more. The type of reaction formed in a chemical calculator is determined by the reactants and the specific algorithm or rules implemented in the calculator.
How Many Grams Of Water In Chemical Reaction Calculator?
To determine the amount of water in a chemical reaction using a calculator, you would need to know the balanced chemical equation and the quantities of the reactants or products involved. By performing stoichiometric calculations, you can convert the given quantities (in moles, mass, or volume) to grams of water. The specific procedure may vary depending on the reaction and the information provided.
