Table of Contents
Here, we will describe the SO2 Molecular Geometry in detail. Sulfur Dioxide which is also recognized as Sulphur Dioxide is the entity of a bond between Sulfur and Oxygen atoms. It can be seen as a formula written as SO2. Here we will provide a description of SO2 molecular geometry, SO2 electron geometry, SO2 bond angle, and SO2 Lewis structure.
SO2 Lewis Structure
Before quickly jumping into the lewis structure of SO2, let’s have an abrupt discussion regarding the usefulness of the lewis structure and the steps to draw it.
Lewis structure is the placement of the electrons around the atoms of a compound. This structure benefits us to know about the kind of bonds and the number of bonds that form the compound.
Now let’s go through the method of drawing lewis structure:
- Step 1 – Figuring out the total number of valence electrons in the molecule is the first and most remarkable step. While doing so, do take care of the +, – signs. A ‘+’ sign implies losing electrons and ‘-‘ means gaining.
- Step 2 – Next thing is determining the central atom. The atom with the highest number of binding locations is the central atom.
- Step 3 – The third step is building a skeleton structure with single bonds only.
- Step 4 – Next, our work is achieving the octet of the atoms with the resting electrons, after the formation of the single bonds. Always start with the electronegative atoms then bring them to the electropositive ones.
- Step 5 – Giving double or triple bonds is fundamental if it is needed for fulfilling the octet rule for all atoms.
- Step 6 – At last, it’s important to analyze if all the atoms are having their lowest possible formal charge.
Formal charge calculation can be done using:-
Formal charge = [no. of valence electrons] – [electrons in lone pairs + 1/2 the number of bonding electrons]
This formula explicitly indicates the relationship between the number of bonding electrons and their link to how many are formally “kept” by the atom.
For example, applying this to BH4 we get:
The number of valence electrons for boron is 3. The number of non-bonded electrons is zero. The total number of bonding electrons around the boron is 8 (full octet). One-half of this is 4.
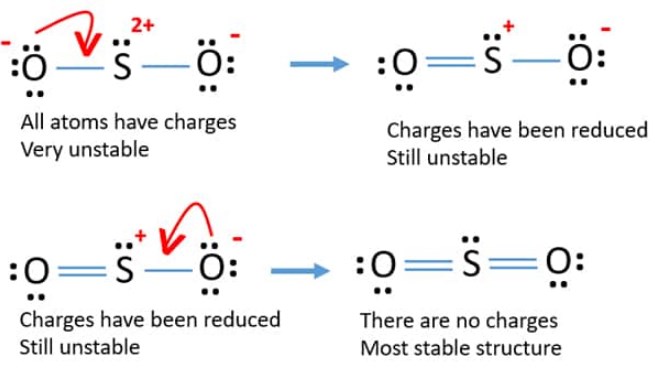
Now let’s see the lewis structure of SO2.
In SO2, the sulfur’s valence electron = 6
And the valence electrons of oxygen = 6
There are 2 oxygen atoms in the compound, thus = 6*2 = 12
So, total valence electrons = 18
After drawing the skeletal structure, we can see that none of the atoms can achieve their octet with single bonds. So there is a need for a double bond. Thus the number of electrons employed in double bonds = 8
Deducting that from the total valence electrons we get 10 electrons remaining. We need to put these remaining electrons around the atoms as per the need.
This will lastly complete the octet of the atoms. Oxygen has 2 lone pairs and sulfur has 1 lone pair.
At last, don’t forget to confirm the formal charge of all the atoms!
The next topic we need to know is the hybridization of SO2.
SO2 Hybridization
The hybridization of SO2 is Sp2.
Now hybridization of SO2 can be discerned in two ways, one is the theory and the 2nd is straight applying the formula. I would advise understanding the theory first and then you can absolutely go for the formula.
A quick edge for you, when 1 s orbital unites with 2 p orbitals it derives in Sp2 hybridization having 3 equivalent orbitals.
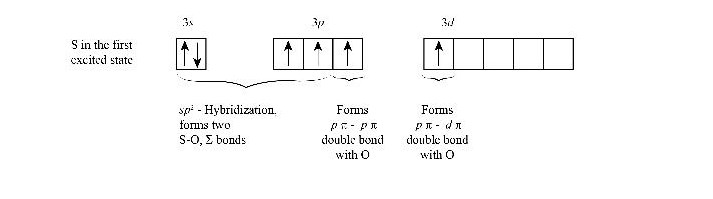
As well, in case of SO2, the ground state electronic configuration is 1s2 2s2 2p6 3s2 3p4. When in an excited state, one electron from 3px, motions to 3d orbital. Thus we have 3p3.
Now, the 3s2 and 3p3 associate to form Sp2 hybridization with 3 equivalent orbitals, containing 2 paired electrons and 2 unpaired.
For forming 2 sigma bonds with oxygen atoms, sulfur needs the 2 unpaired electrons from the Sp2 hybridized orbitals. And the resting 2 paired orbitals form the lone pair of sulfur.
Thinking about the other 2 electrons of 3p which were not associated with hybridization?
Well, those two (i.e one of the 3p orbital and another electron in 3d) formed the ? bonds between sulfur and oxygen. There’s an image attached below for better understanding.
Now coming to the formula part.
The formula for finding hybridization of any compound is;
H = ½ [ V+M-C+A]
Where,
- H depicts Hybridization
- V is the no. of valence electrons
- M is the count of monovalent atoms present
- C represents the cationic charge
- A represents the anionic charge
Here, if H is 2, it’s Sp hybridization.
When, H = 3, it’s Sp2 hybridization.
When, H = 4, it’s Sp3 hybridization.
By the same token H = 5, its Sp4 hybridization.
And finally, when, H is 6, it will be Sp3d2 hybridization.
For SO2, the number of valence electrons of the S atom = 6 and the number of monovalent atoms = 0, because oxygen is a divalent atom.
Here cationic and anionic charges will be 0 as it’s a neutral compound.
Thus, H = ½ [6+0-0+0]
H = ½ * 6
H = 3 = Sp2 hybridization.
I think the hybridization of SO2 is clear from both the explained concepts.
SO2 Bond Angle
The SO2 has a bond angle of 120-degree. One single atom of Sulphur is bonded with two atoms of Oxygen covalently. It causes a repulsion of electron pairs to form the 120-degree angle.
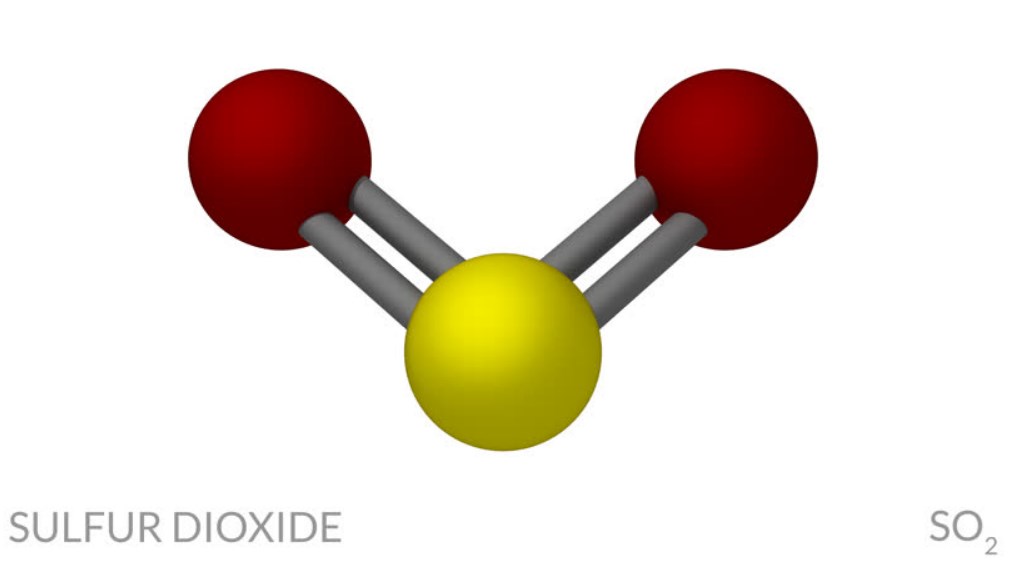
SO2 Molecular geometry
The molecular geometry of SO2 is bent, with a bond angle of 120°.
We can easily find out the molecular geometry of any compound using the given chart.
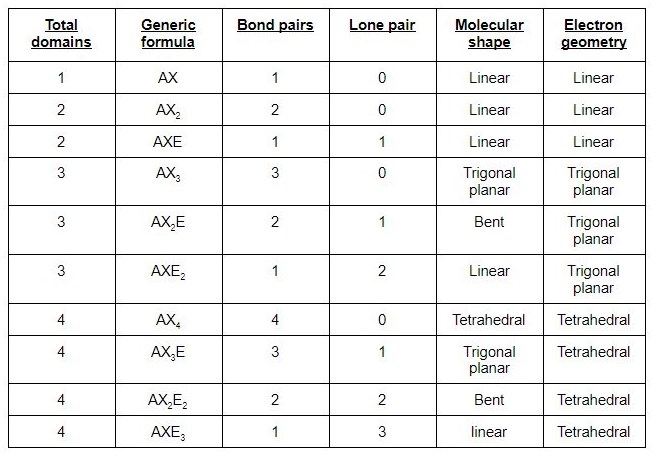
Here, A = central atom, X = surrounding atoms and E = the lone pairs. SO2 is an AX2E type molecule, with 2 surrounding atoms i.e oxygen, and 1 lone pair of sulfur.
But the electron geometry of SO2 is trigonal planar. You must be wondering about this new term, right? Let me explain.
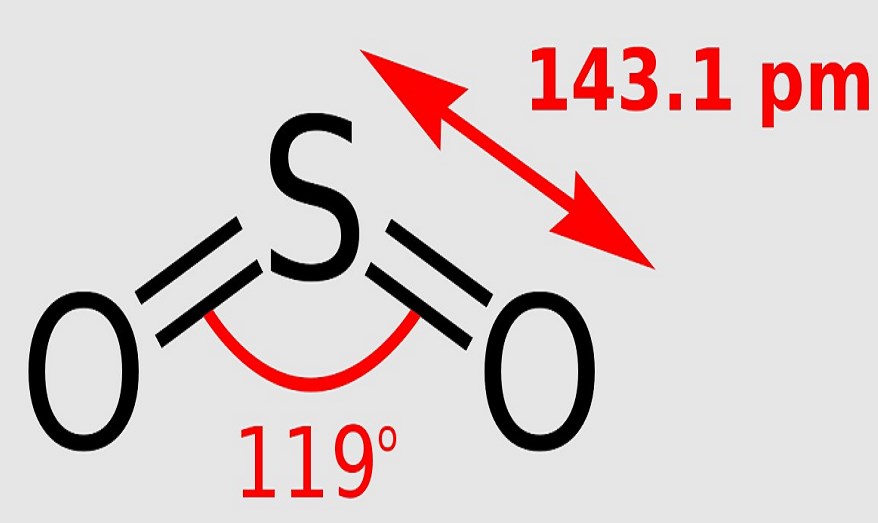
So, electron geometry is different from molecular geometry because it considers all the electron pairs (including lone pairs) while determining the shape. Whereas molecular geometry considers only the atoms.
In absence of a lone pair, both the geometries are the same for any compound.
Below is the 3D view of the geometry of the SO2 molecule.
Now let’s learn the last topic of this article, the molecular orbital diagram of SO2.
SO2 Molecular Orbital Diagram
The molecular orbital diagram of SO2 is attached below:
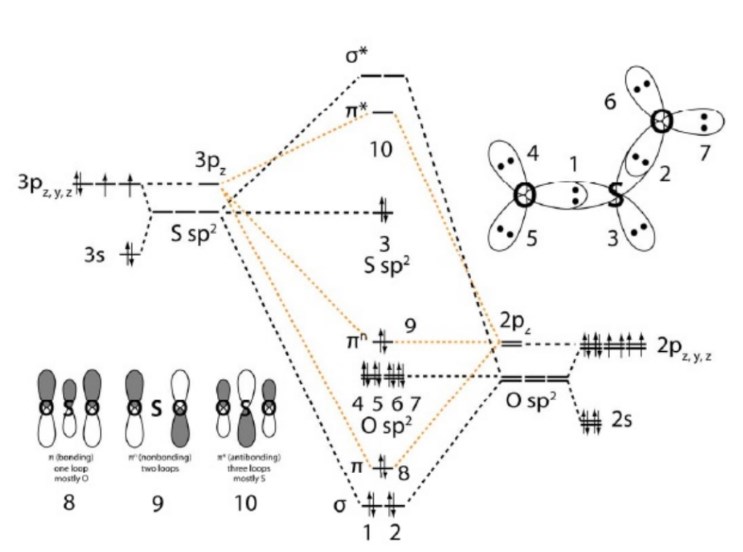
A molecular orbital diagram gives us an idea about how the atomic orbitals of two different atoms can fuse and give rise to a new orbital.
This further helps us to find out the bond order, bond length, and bond strength of any compound.
In this MO we can see that the AO of sulfur, which is on the left-hand side associates with the AO of oxygen on the right-hand side.
We can see 18 electrons are filled in the orbitals with the proper rule.
There are certain non-bonding orbitals present there as well. Also, the antibonding orbitals are vacant in the situation of SO2.
This adds up to the explanation of the molecular orbital diagram of SO2.
Similarities between Sulfur and Oxygen atoms
- Both O and S have the same outer electric configuration of ns2 and np4.
- O and S are usually divalent.
- O and S are non-metals.
- Both exhibit allopatric form.
- In reaction with metals, both react with the oxidation state of -2.
- While reacting with nonmetals, both form covalent compounds, for instance, H2O, H2S, CO2, and CS2.
Dissimilarities between oxygen and sulfur
- Oxygen has two allotropic forms while Sulphur has 3 allotropic forms.
- Oxygen is gas at ordinary temperature while Sulphur is solid at ordinary temperature.
- Oxygen is sparingly soluble in water while Sulphur is not soluble in water.
- Oxygen helps in combustion while Sulphur is combustible itself.
- Oxygen is paramagnetic in nature while Sulphur is diamagnetic in nature.
- Oxygen does not react with water while when steam is passed through boiling sulfur, a little hydrogen sulfide and sulfur dioxide are made.
- Oxygen does not react with acids while Sulphur is readily oxidized by concentrated sulfuric acid or nitric acid.
SO2 Preparation
SO2 can be produced in several methods. I am breaking down each procedure to make it easy to understand!
- Method 1 – The main production of SO2 is during the manufacture of sulfuric acid using the contact process. Among all other ways to manufacture SO2, this method is widely used in industries. ( Because Chemistry has a lot to do with history, here’s a historical fact for you! In 1979, the United States used 23.6 million tonnes of SO2 for manufacturing sulfuric acid.)
- Method 2 – SO2 can be produced by burning sulfur or materials containing sulfur.
S + O2 —> SO2
2 H2S + 3O2 —> 2H2O + 2SO2
- Method 3 – SO2 production can also be done by the roasting of pyrite, sphalerite, and cinnabar ( sulfide ores).
- Method 4 – In the formation of calcium silicate cement, SO2 is produced as a byproduct.
2 CaSO4 + 2SiO2 + C —> 2CaSiO3 + 2SO2 + CO2
- Method 5 – In the laboratory, the reaction between hot concentrated sulfuric acid and copper turnings leads to SO2 formation.
Cu + 2H2SO4 —> CuSO4 + SO2 + 2H2O
- Method 6 – Natural calamities like volcanic eruptions can produce a large amount of SO2.
S02 Is Polar or Nonpolar?
Sulfur dioxide is polar in nature. The difference in electronegativity between sulfur and oxygen atoms creates polarity in the molecule. Oxygen has a greater electronegative potential than sulfur. Therefore, oxygen exerts more pull on the covalent bonds in sulfur dioxide. The portion of the molecule that has both oxygen atoms on it is slightly negatively charged.
Whereas, the portion which has the sulfur atom has a slightly positive charge. This makes SO2 a polar molecule like H2S. In addition, the unbonded electrons on the sulfur and oxygen create repulsion between atoms.
This is another cause of the polarity of the sulfur dioxide molecule.
Sulfur dioxide Effects on Humans
- Sulfur dioxide is a toxic gas and is directly harmful to human health.
- It can irritate the skin and mucous membranes of the eyes, nose, throat, and lungs.
- Its high concentrations can cause inflammation and irritation of the respiratory system.
- High emissions of sulfur dioxide in the air can lead to the formation of other sulfur oxides (SOx).
- SOx can react with other compounds in the atmosphere to form small particles.
- These small particles may penetrate deeply into the lungs and their sufficient quantity can contribute to health problems.
Conclusion
- The molecular geometry of sulfur dioxide is a bent shape.
- Sulfur to the Oxygen ratio in Sulfur dioxide is 1:2.
- Sulfur dioxide molecule has two double bonds between the Sulfur atom and Oxygen atoms.
- There are 5 lone pairs of electrons in the molecule of SO2.
- Molar mass of sulfur dioxide = 64.066 g/mol.
- SO2 gives a week acid solution when dissolved in water.
This article explains pretty much everything you need to know about SO2. Before studying the reactions and equations involving SO2, go through this article to clear all your concepts regarding lewis’s structure, geometry, hybridization, and MO diagram of SO2.
If you have any doubt feel free to contact us anytime. Have a happy reading!
